Week of April 17, 2026
The Week at a Glance:
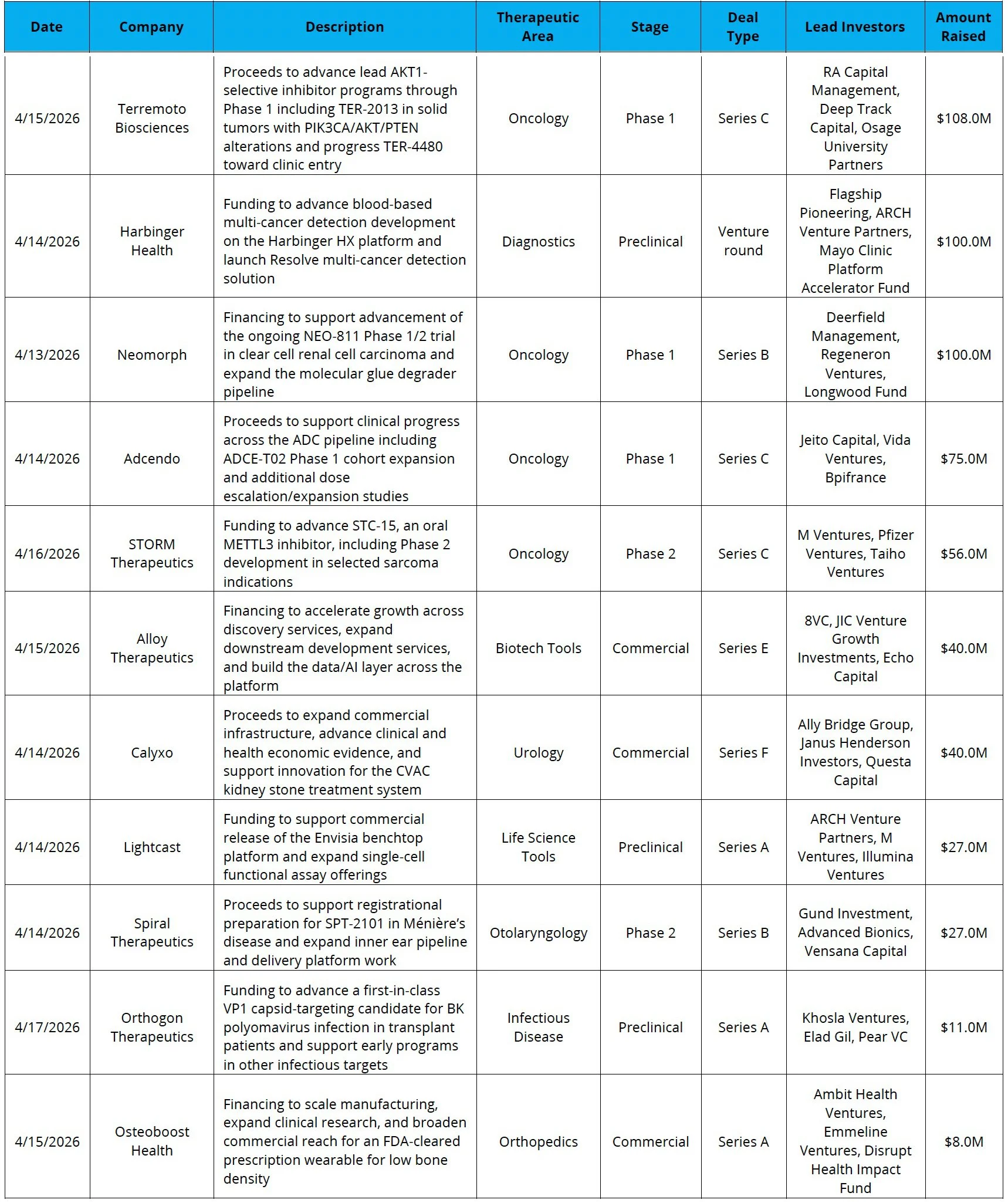
Obesity Is Still in Vogue: Kailera Therapeutics, a company developing GLP-1 therapies for obesity, raised $625.0M in its IPO, making it one of the largest biotech IPOs in history
It’s Not Only Kailera: Alamar Biosciences raised $191.0M in its IPO at the top of its $15 to $17 per share range, while Mobia Medical and Odyssey Therapeutics have each filed to go public for up to $100.0M
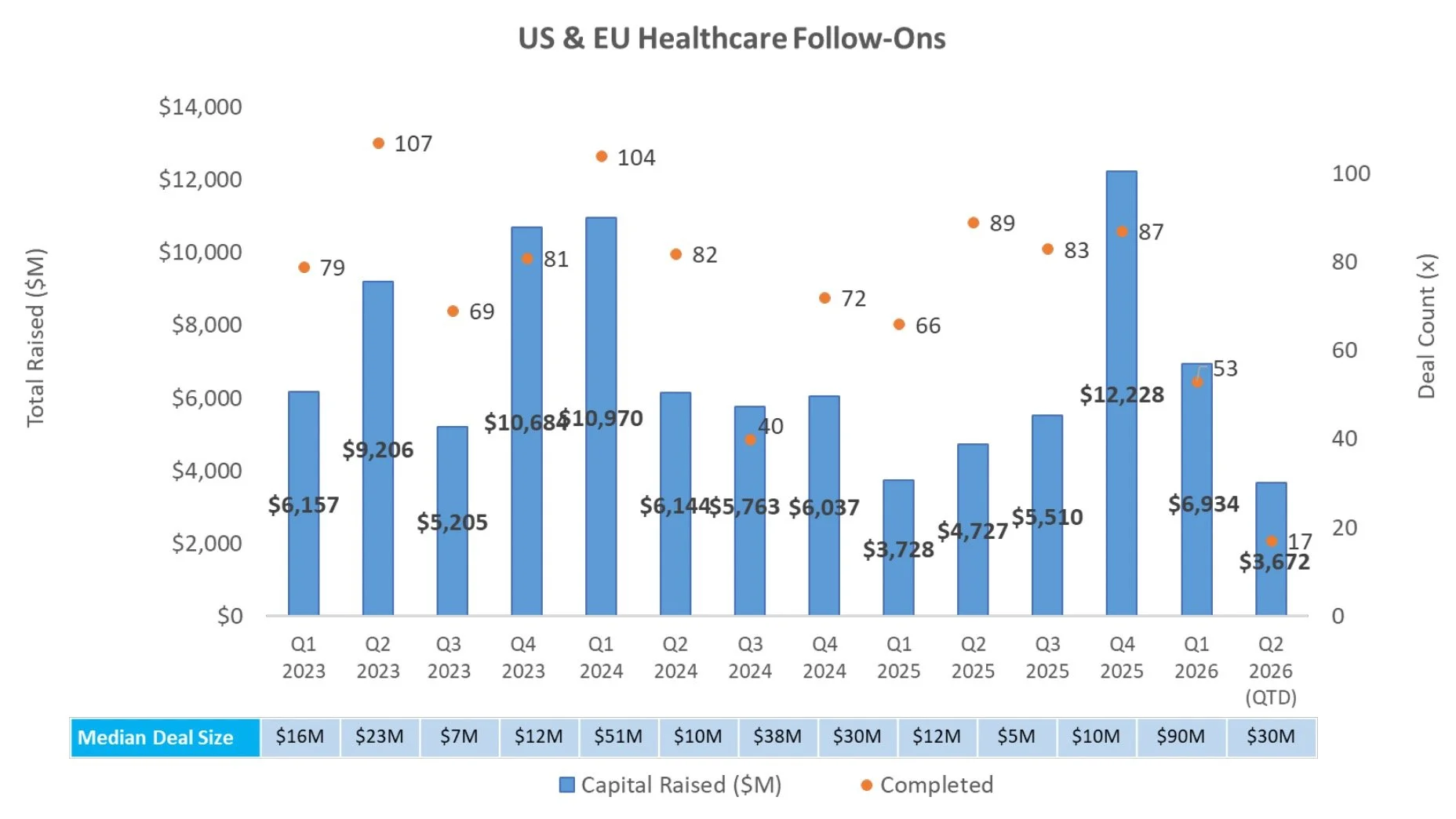
Follow-On Activity Remains Strong: In addition to Revolution Medicines’ offering, Spyre Therapeutics and Allogene Therapeutics closed follow-on offerings of $463.5M and $200.4M, respectively, while Trevi Therapeutics and MeiraGTx announced offerings of $150.0M and $100.0M, respectively
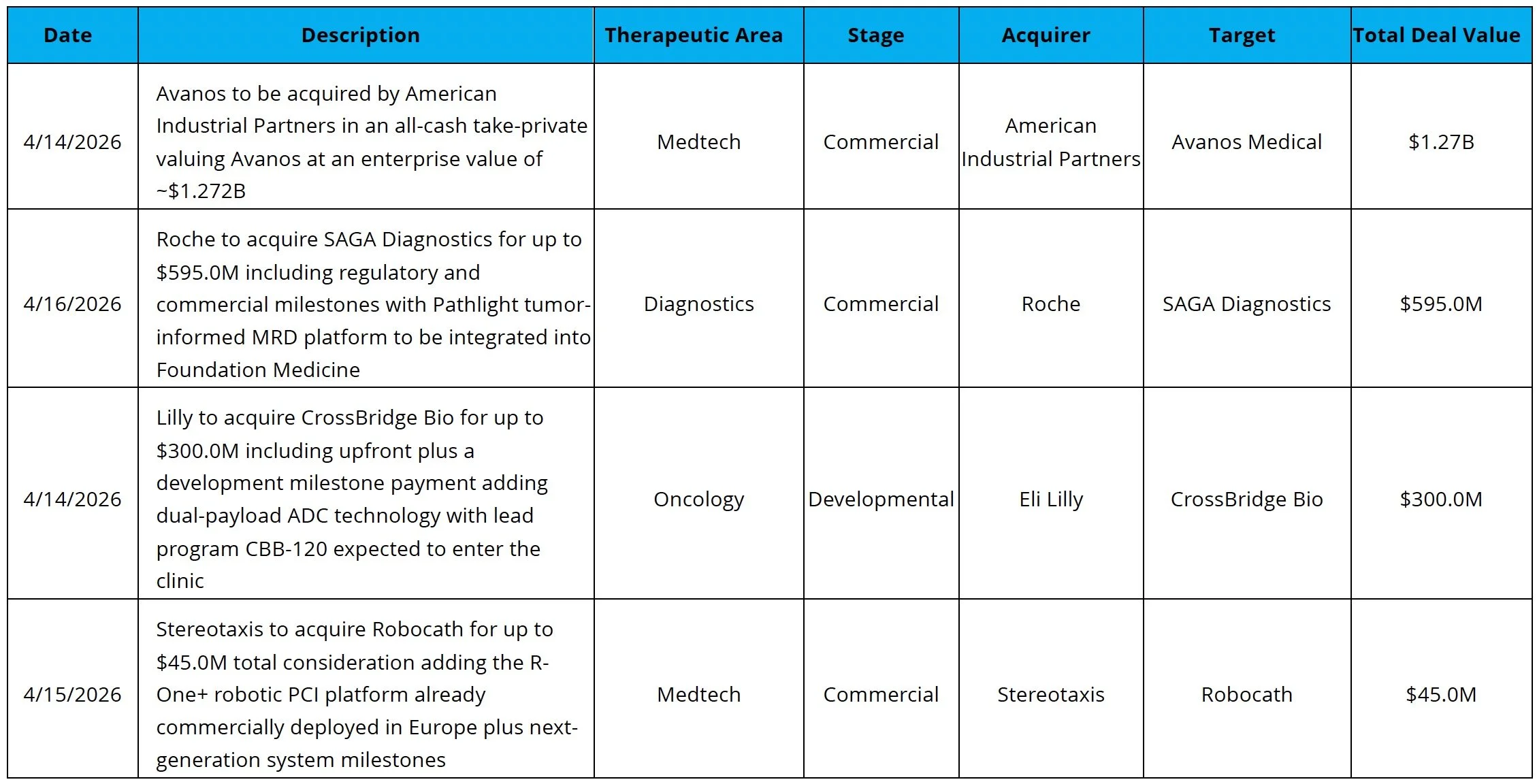
UCB Expands Within Epilepsy: UCB acquired Neurona Therapeutics for up to $1.2B, expanding its epilepsy franchise through Neurona’s focus on drug-resistant mesial temporal lobe epilepsy
Pancreatic Cancer Data Impresses: Revolution Medicines raised $2.2B after reporting paradigm-shifting data from its Phase 3 RASolute trial in metastatic pancreatic cancer, showing that its pan-RAS(ON) inhibitor reduced the risk of death by 60% versus chemotherapy in the 2L setting
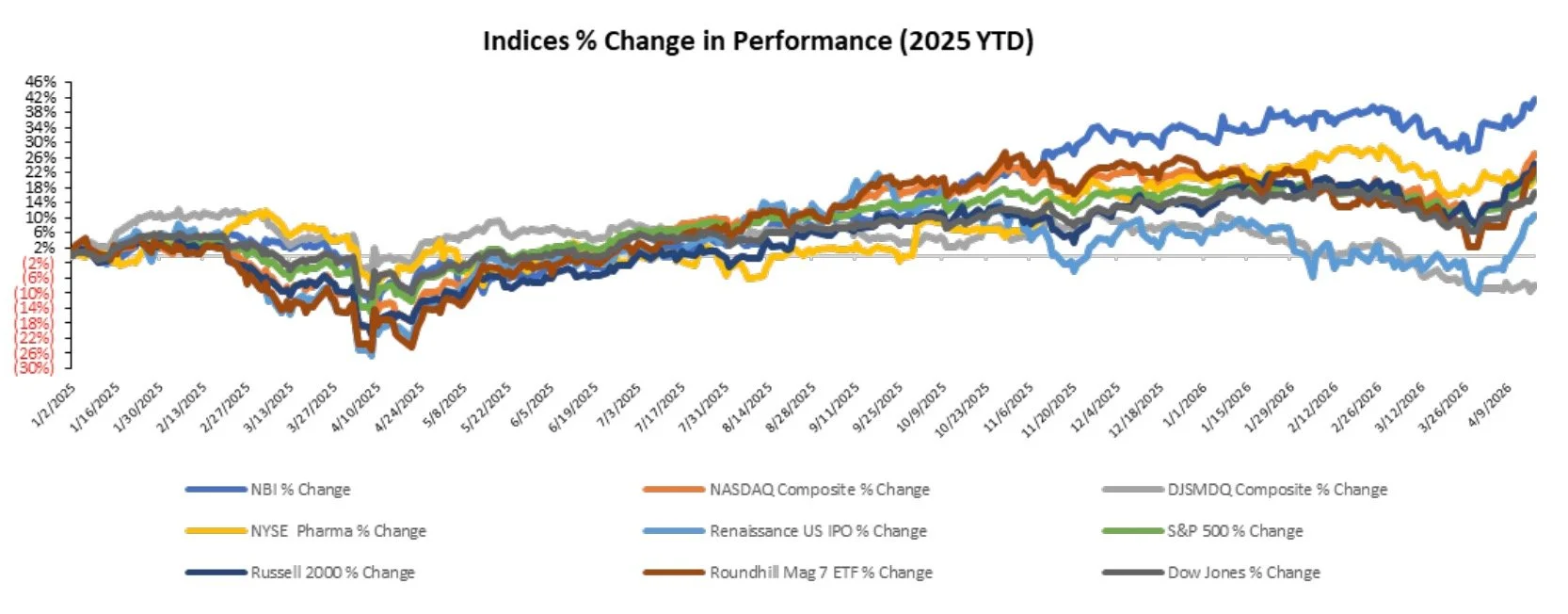
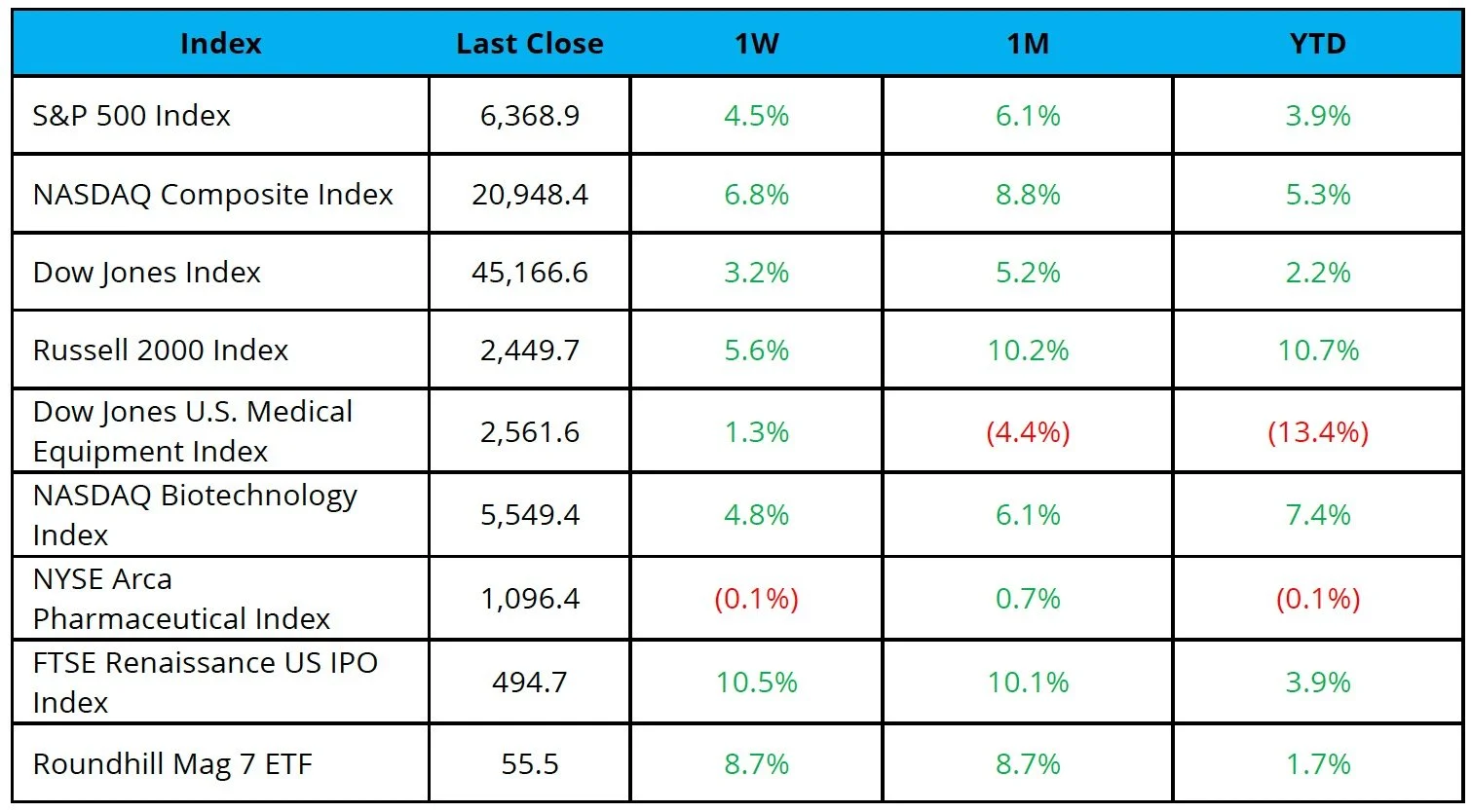
Markets Overview
The S&P 500, Nasdaq, and Dow were up 4.5%, 6.8%, and 3.2%, respectively, over the last week
Markets grew increasingly optimistic on signs of de-escalation in the Iran conflict; however, reports that the US seized an Iranian-flagged ship suggest tensions may remain elevated
The NYSE Pharma Index was down 0.1%, while the NBI was up 4.8% over the last week
Notable changes in share price:
Revolution Medicines (NASDAQ: RVMD): Shares jumped 54.1% after the Company reported positive results from its Phase 3 RASolute 302 trial in patients with metastatic pancreatic cancer and announced a $2.2B stock offering and convertible senior notes pricing
Adlai Nortye (NASDAQ: ANL): Shares rose 31.1% following Revolution Medicines’ readout, as Adlai is developing a similar but potentially differentiated pan-RAS(ON) inhibitor. Adlai also raised a $150.0M PIPE, capitalizing on growing interest in the pan-RAS(ON) space
Replimune (NASDAQ: REPL): Shares fell 54.4% after the FDA issued their second CRL for Replimune’s melanoma drug
Sources: Pitchbook, Biomedtracker, and CapIQ
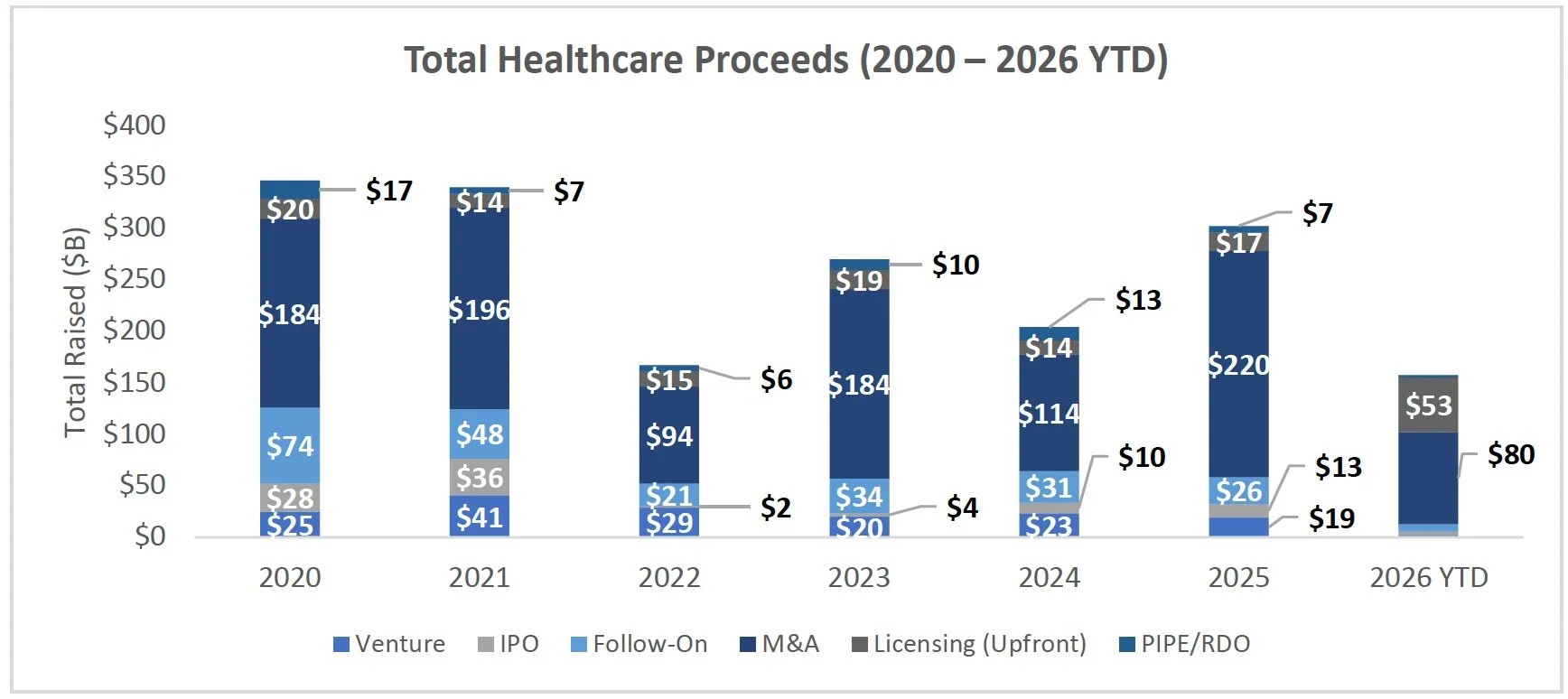
Equity Markets
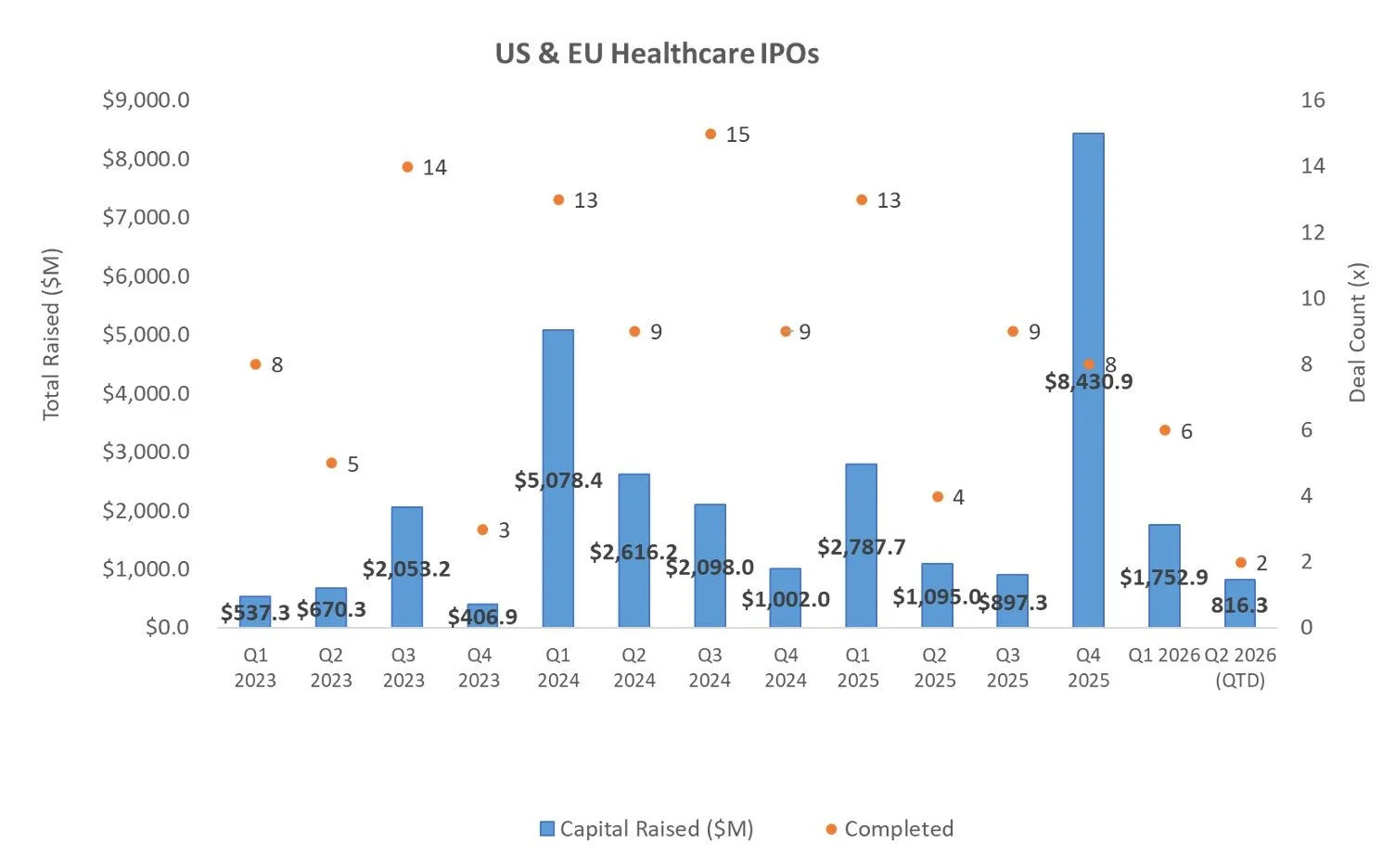
IPO
Two companies filed an S1 last week:
Mobia Medical plans to raise up to $100.0M to support commercialization of its vagus nerve stimulation system for ischemic stroke survivors with moderate to severe upper extremity impairment
Odyssey Therapeutics plans to raise up to $100.0M to support development of its Phase 2 oral RIPK2 small molecule for ulcerative colitis and Crohn’s disease, as well as its preclinical SLC15A4 small molecule candidate
All 24 additional companies in the IPO queue are pursuing raises below $40.0M
IPOs priced in 2025 and 2026 have generated a median and average return of (6.5%) and 5.2% YTD, respectively. Half of newly public companies are trading above their offer price
After-Market Performance by Stage
Clinical-stage after-market performance (N=18): 23.2% (average), 1.9% (median)
Commercial-stage after-market performance (N=24): (7.4%) (average), (12.3%) (median)
After-Market Performance by Sector
Biopharma (N=18): 23.2% (average), 1.9% (median)
MedTech (N=23): (13.9%) (average), (18.1%) (median)
Source: CapIQ
Follow-On Offering Markets:
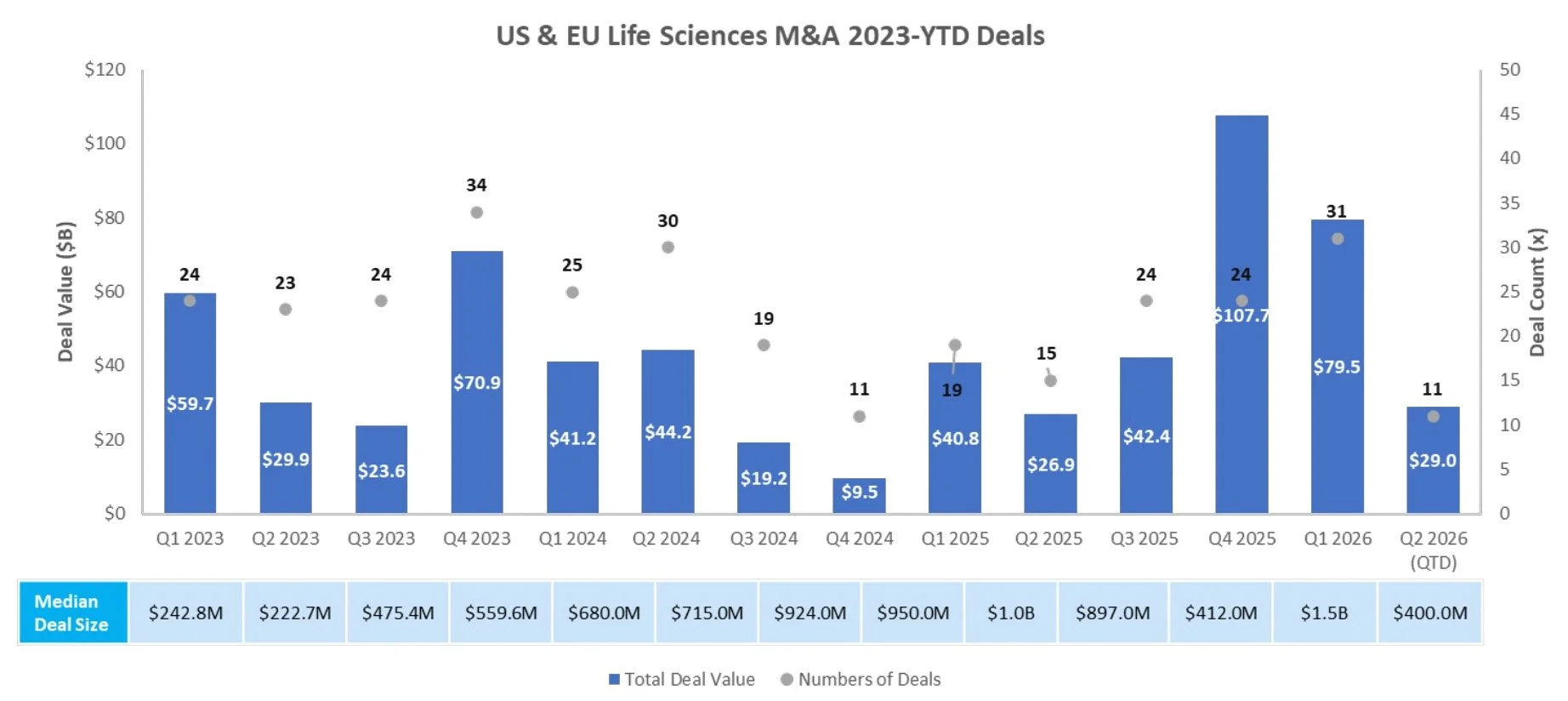
There were six follow-on equity offerings totaling $2.6B last week, including:
Revolution Medicines (NASDAQ: RVMD) closed a $1,725.0M public offering to fund the development of its clinical stage asset daraxonrasib (RMC-6236), a RAS(ON) inhibitor for RAS-addicted cancers
Spyre Therapeutics (NASDAQ: SYRE) closed a pricing of a $463.5M public offering to fund the development of its clinical stage assets including SPY001, an anti-α4β7 monoclonal antibody for inflammatory bowel disease (ulcerative colitis and Crohn’s disease)
Allogene Therapeutics (NASDAQ: ALLO) closed a pricing of a $200.4M public offering to fund the development of its clinical stage asset cema-cel, an allogeneic anti-CD19 CAR-T for large B-cell lymphoma
Trevi Therapeutics (NASDAQ: TRVI) announced a pricing of a $150.0M public offering to fund the development of its clinical stage asset Haduvio (nalbuphine ER) for chronic cough in idiopathic pulmonary fibrosis and other ILD / refractory chronic cough
MeiraGTx Holdings (NASDAQ: MGTX) announced a pricing of a $100.0M public offering to fund development of its clinical stage genetic medicines pipeline including AAV-AQP1 for radiation-induced xerostomia and other gene therapy programs
Source: Biomedtracker
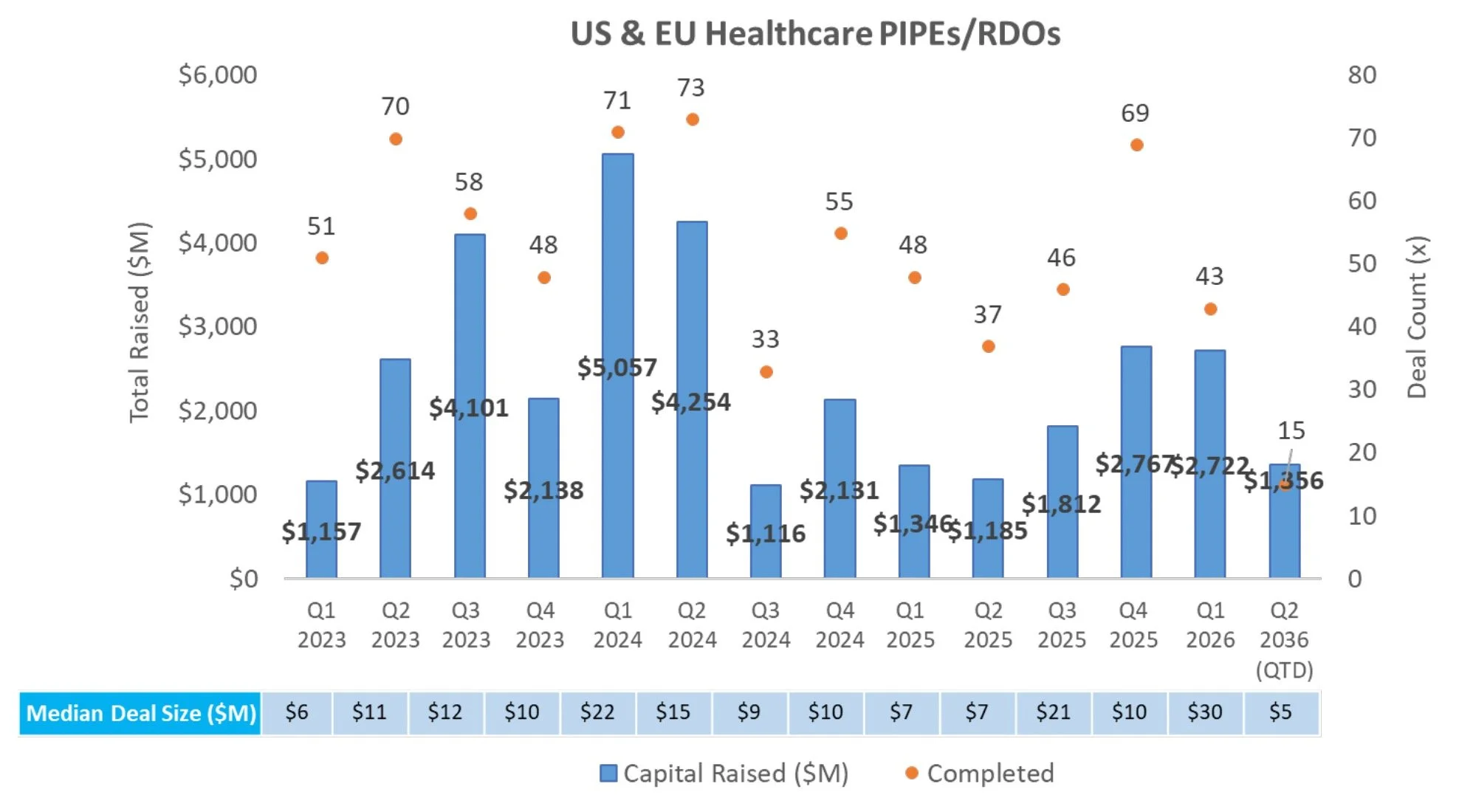
PIPE/RDO Markets:
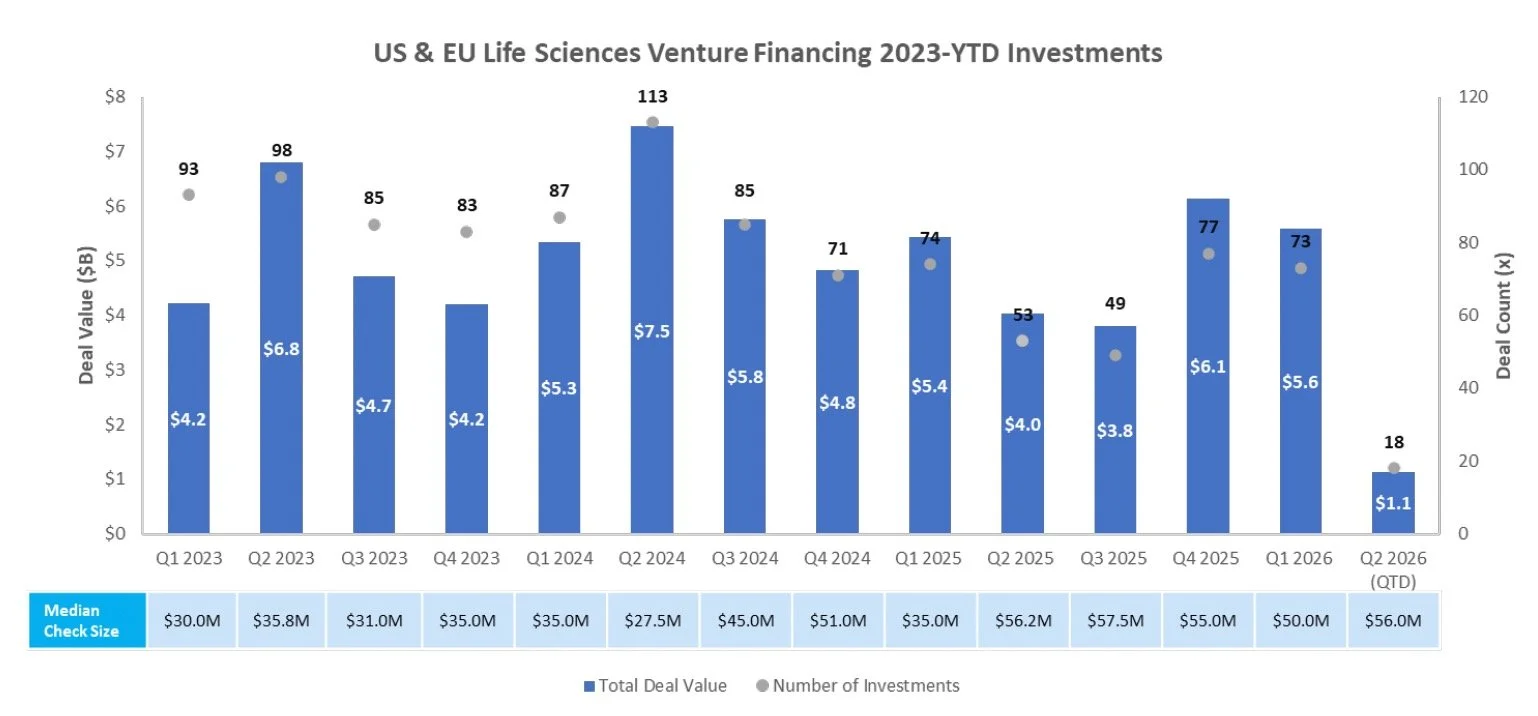
There were eight PIPEs/RDOs last week raising an aggregate of $597.8M
Achieve Life Sciences (NASDAQ: ACHV) announced a $354.0M Private Investment in Public Equity to fund a Phase 3 clinical trial of cytisinicline for e-cigarette cessation and support commercialization of cytisinicline
Obsidian Therapeutics announced a $350.0M Private Investment in Public Equity concurrent with its reverse merger with Galera to advance OBX-115, a genetically engineered autologous TIL therapy in Phase 2 for advanced melanoma and Phase 1 for NSCLC
Adlai Nortye (NASDAQ: ANL) announced a $150.0M Private Investment in Public Equity to fund continued development of its oncology pipeline, including immunotherapies and RAS-targeting programs
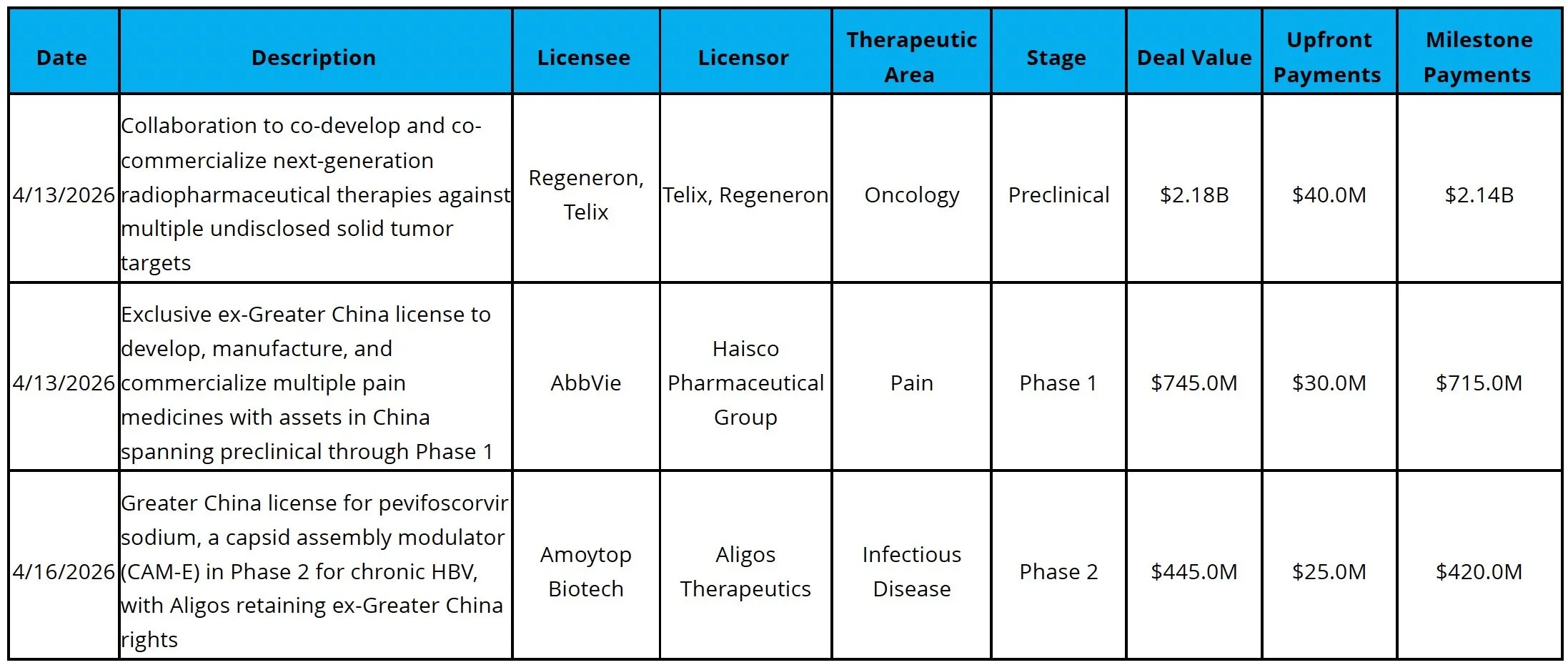
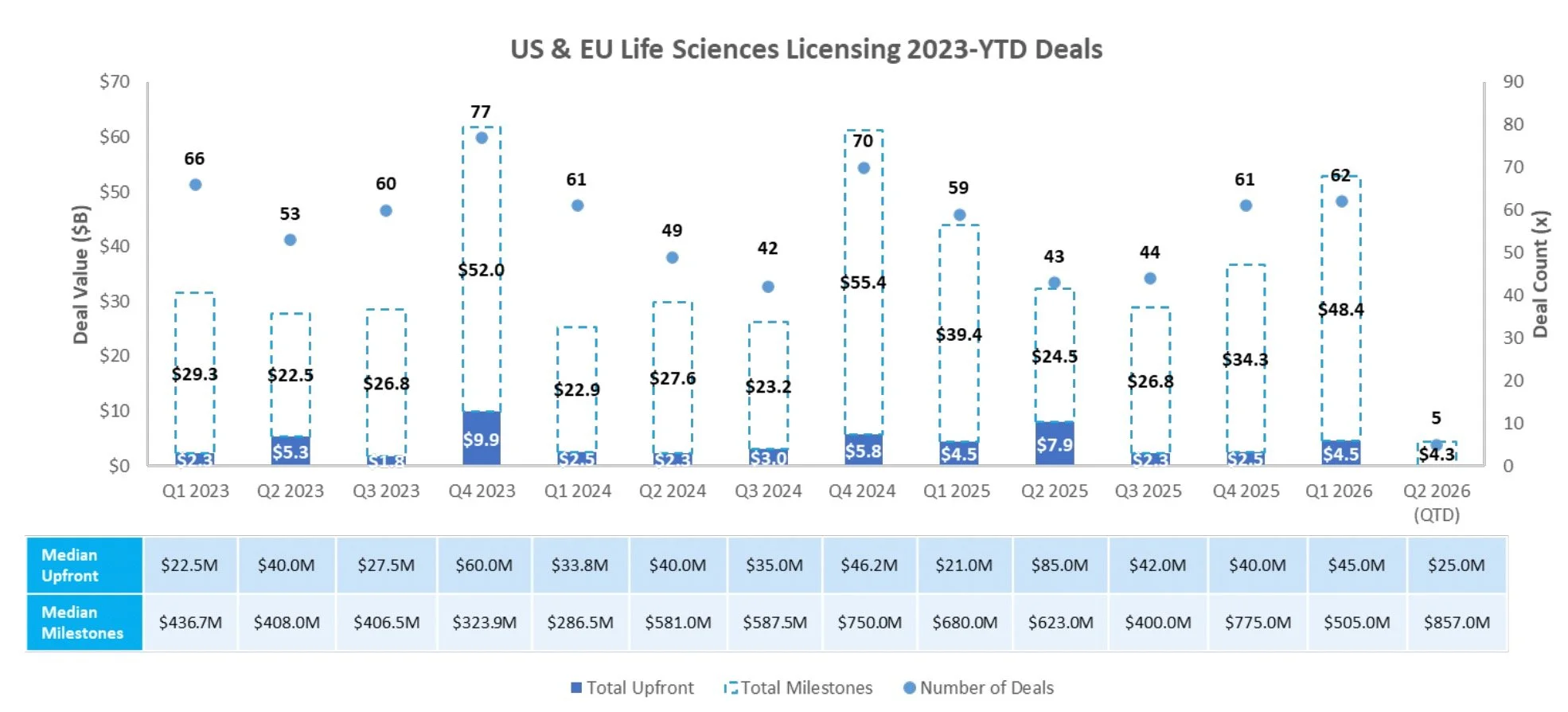
Licensing
Sources: Pitchbook, Biomedtracker, and CapIQ
Sources: Pitchbook, Biomedtracker, and CapIQ
M & A
Venture Financing
Sources: Pitchbook, Biomedtracker, and CapIQ
A MAP to the Future of Targeted Oncology
The latest in our series of healthcare analyst reports focuses on the mitogen-activated protein kinase (MAPK) pathway, one of the most commonly perturbed signaling pathways in human cancer. Flowing from RAS to RAF to MEK to EKR, the pathway is a master regulator of cell growth and survival. Therefore, the amplification of proteins or mutation of key signaling domains are a common hallmark of cancer.
HEALTHCARE MARKET REPORTS ARCHIVE
About DNB Carnegie | Back Bay
DNB Carnegie Back Bay drives global healthcare growth and innovation by providing a full range of strategic advisory and financing capabilities along the continuum of life science and healthcare company development. The DNB Carnegie Back Bay Healthcare Partnership is a marketing term referring to a strategic agreement between DNB Markets, Inc. and Back Bay Life Science Advisors. More information about the DNB Carnegie Back Bay Healthcare Partnership can be found here.
Securities products and services are offered in the U.S. through DNB Carnegie, Inc., a US-registered broker-dealer and a separately incorporated subsidiary of DNB Bank ASA. DNB Carnegie, Inc. is a member of the Financial Industry Regulatory Authority (“FINRA”) and the Securities Investor Protection Corporation (“SIPC”). Securities products and services are offered in the European Economic Area through DNB Carnegie.