Week of May 15, 2026
The Week at a Glance:
FDA Drama with Makary’s Departure: FDA Commissioner Marty Makary resigned amid reported White House disagreements over flavored e-cigarette approvals, triggering a broader FDA leadership shake-up that included the departure of CDER’s acting director
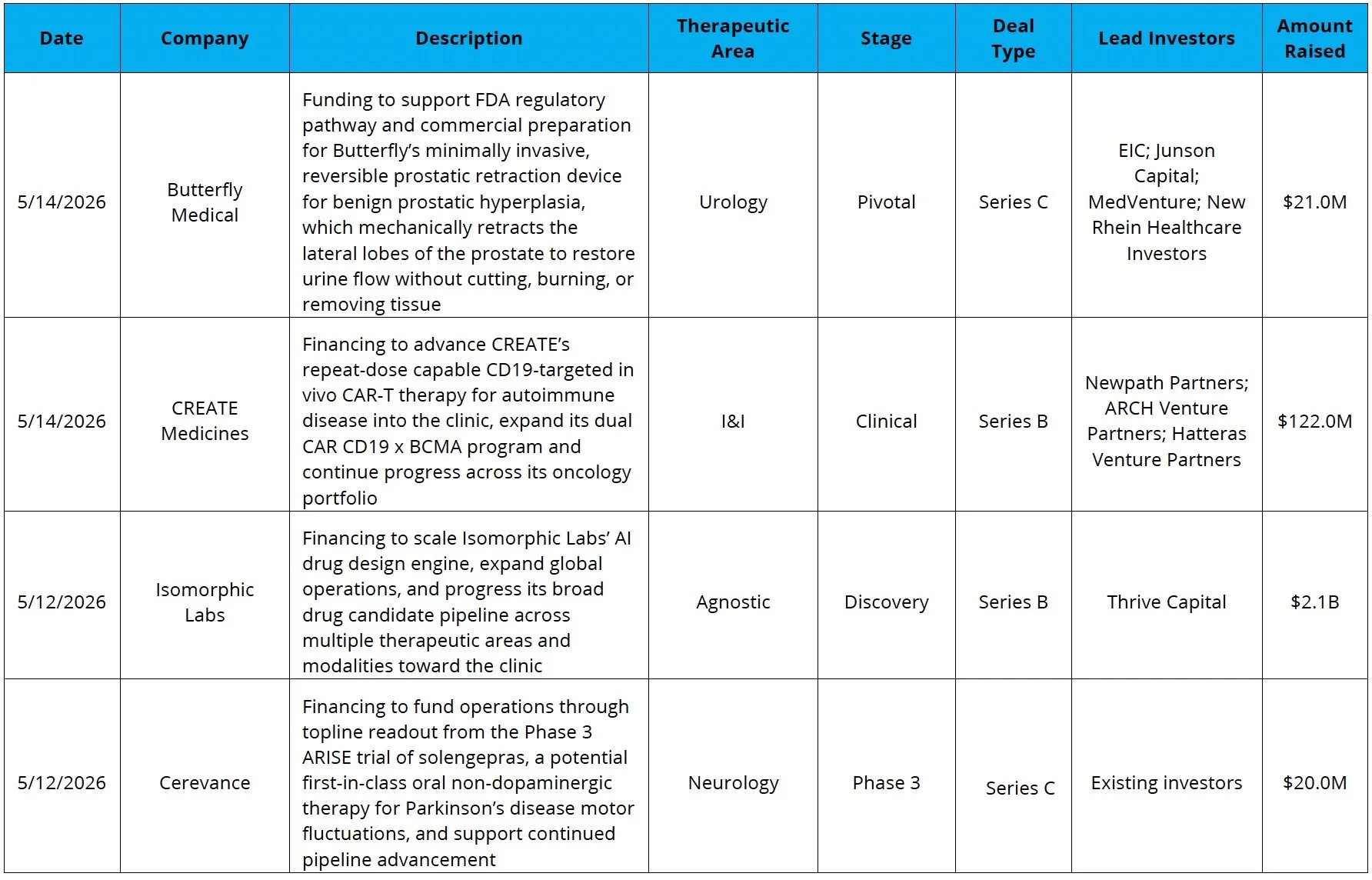
The Second Largest Biotech Private Funding Round Ever: Isomorphic Labs, a DeepMind spin-off focused on AI-driven drug discovery, raised $2.1B in the second-largest biotech funding round to date, trailing only Altos Labs, another AI-enabled drug discovery company that raised $3B in 2022
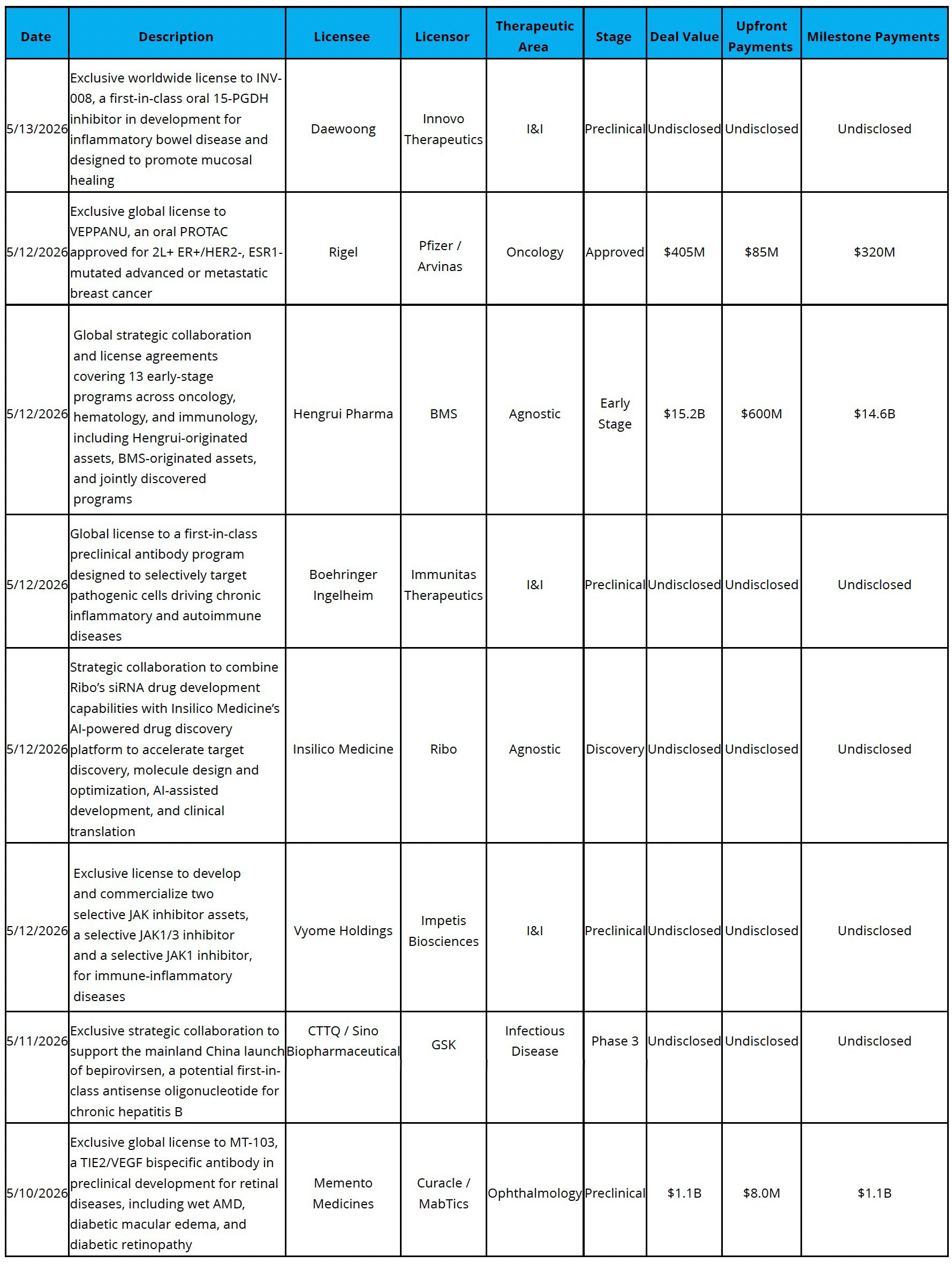
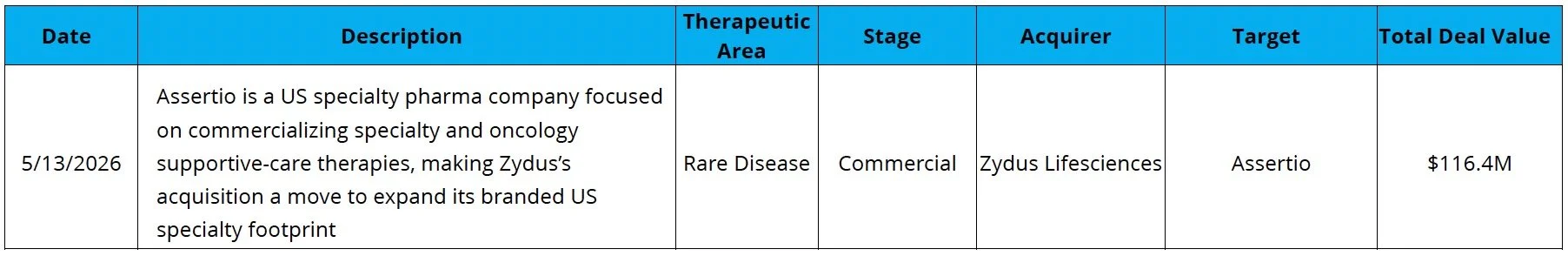
Big Pharma Turning to China: BMS entered a transaction with Hengrui worth up to $15.2B for 13 programs, including eight oncology or immunology assets and five additional programs to be jointly discovered
Markets Overview
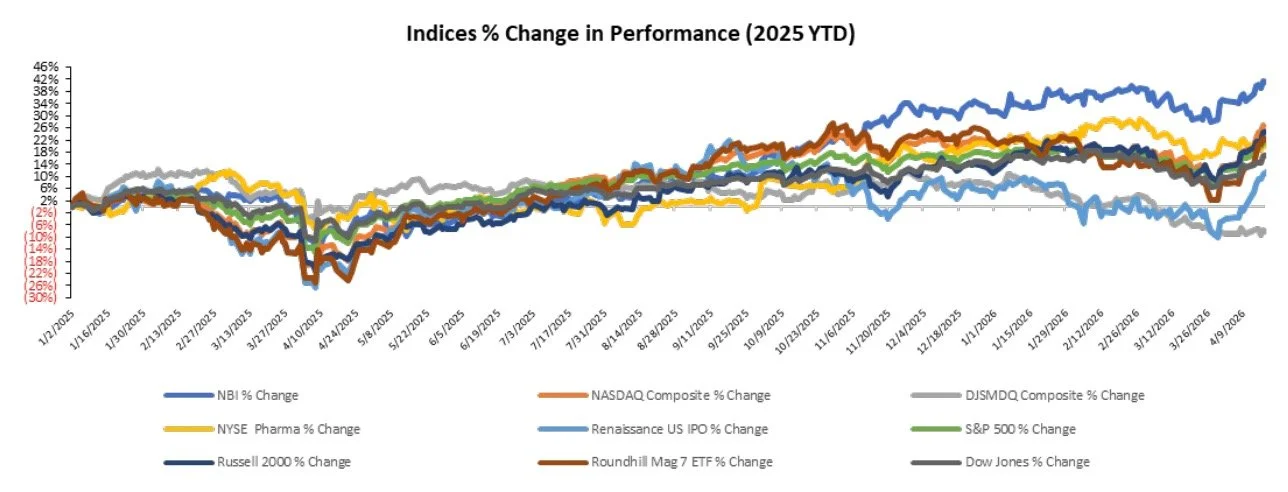
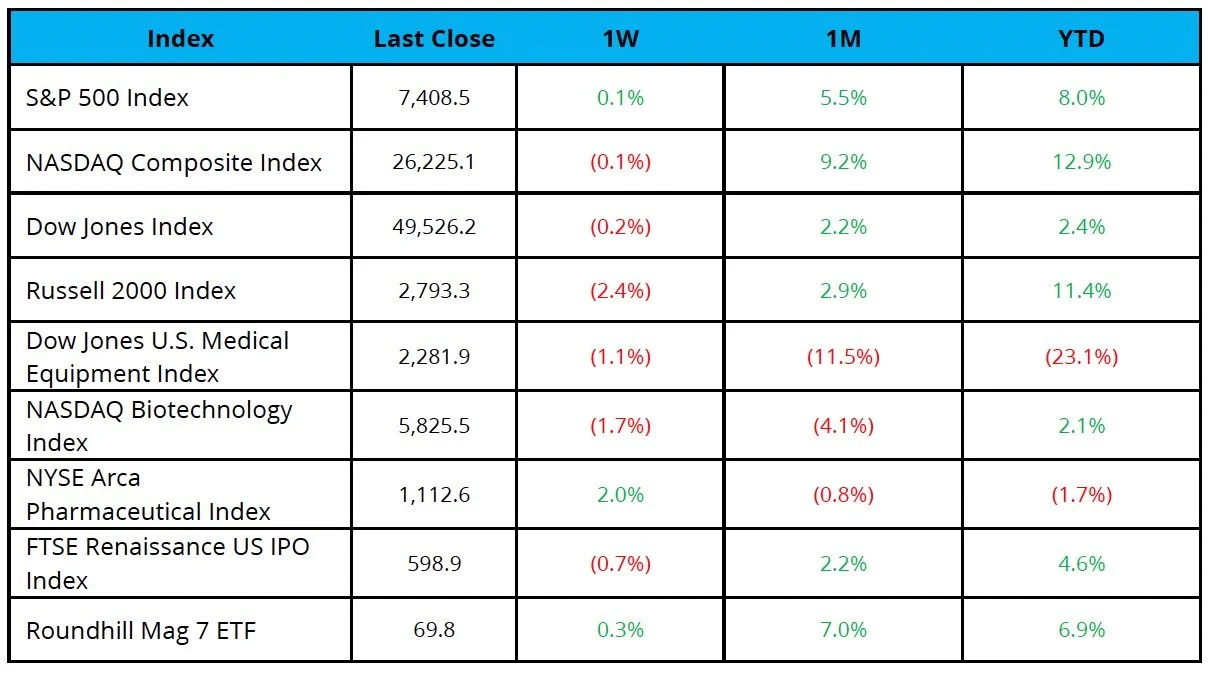
The S&P 500 was up 0.1%, while the Nasdaq and Dow were down 0.1% and 0.2%, respectively, over the last week
Although equity markets were largely flat for the week, Treasuries sold off as bond investors grew increasingly concerned about inflationary pressures stemming from rising energy prices
The NYSE Pharma Index was up 2.0%, while the NBI was down 1.7% over the last week
Notable changes in share price:
Regenxbio (NASDAQ: RGNX): Shares fell 46.6% after the Company reported data from its DMD gene therapy program that revealed serious side effects, making it yet another DMD gene therapy to face safety challenges
Sources: Pitchbook, Biomedtracker, and CapIQ
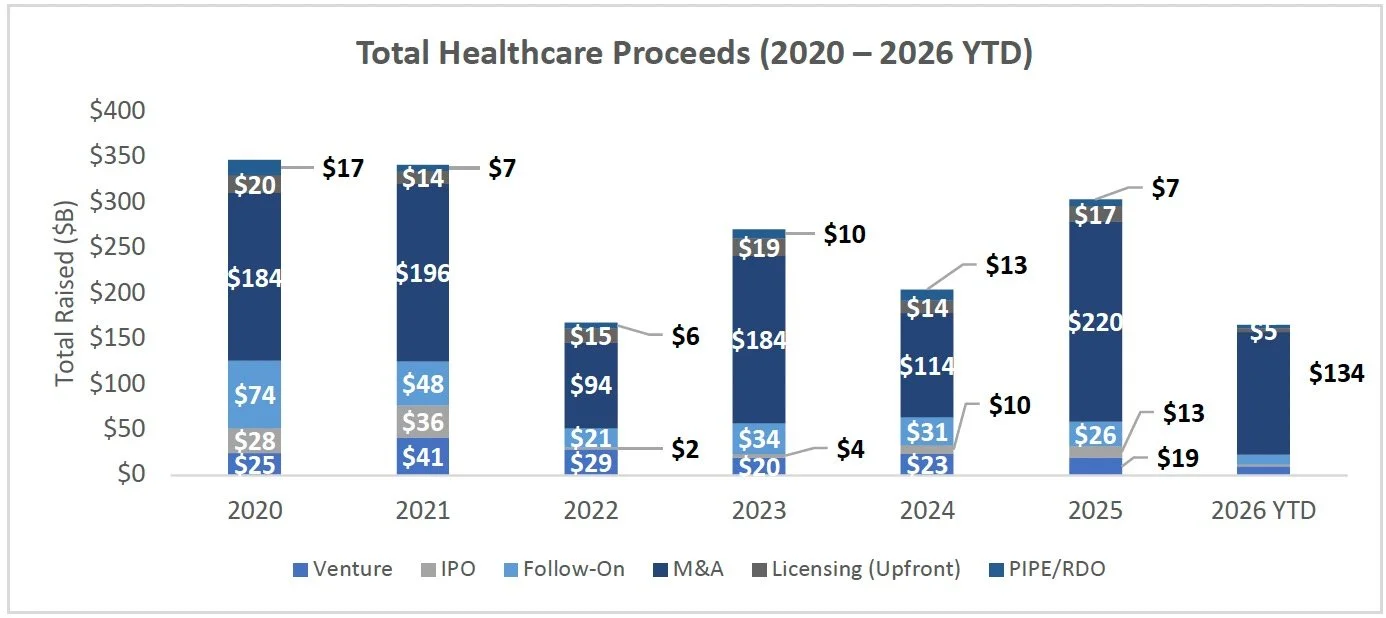
Equity Markets
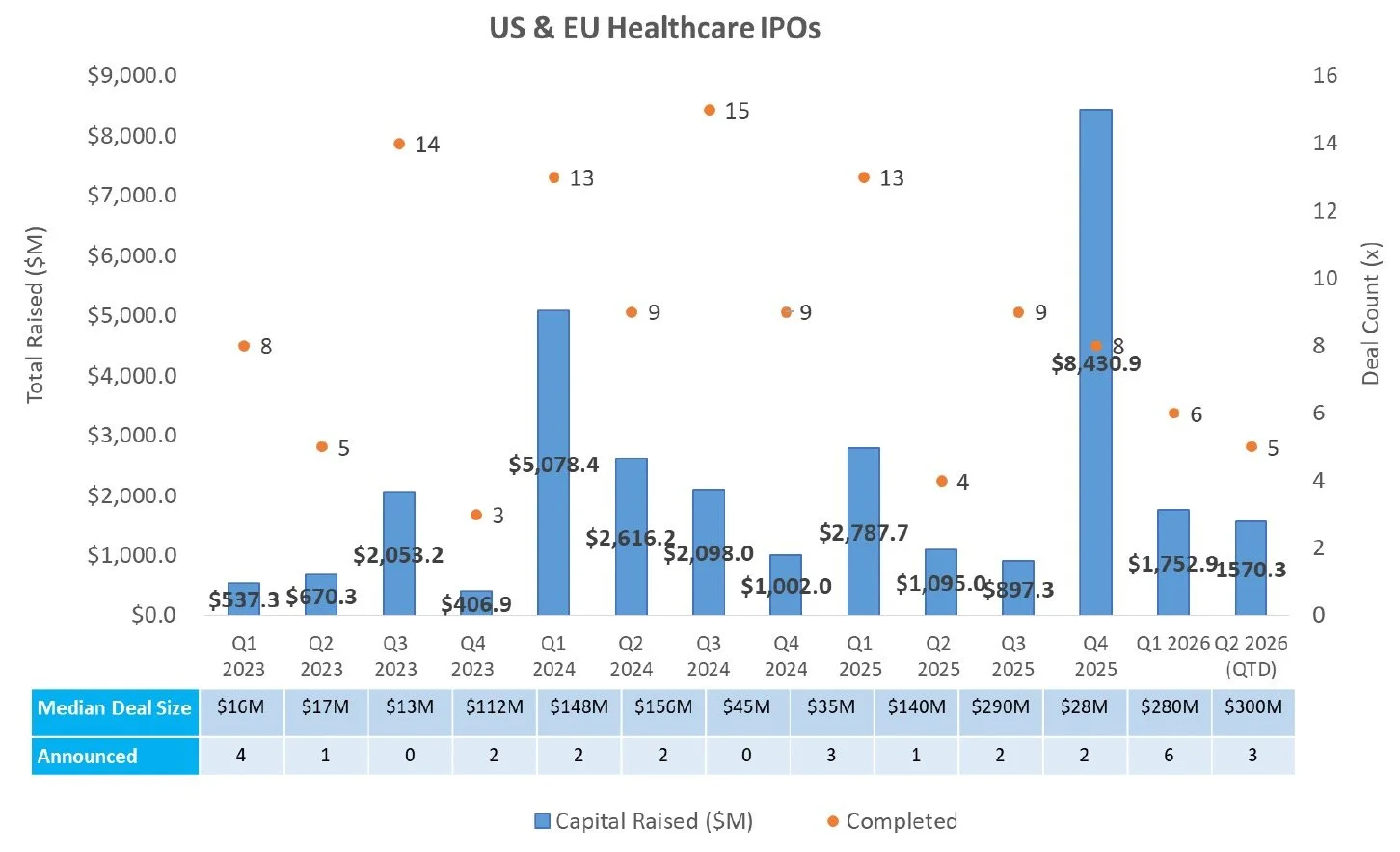
IPO
There were no new S-1s filed last week.
IPOs priced in 2025 and 2026 have generated a median and average return of 6.4% and 18.3% YTD, respectively.
After-Market Performance by Stage
Clinical-stage after-market performance (N=18): 46.3% (average), 31.5% (median)
Commercial-stage after-market performance (N=24): (4.4%) (average), (6.5%) (median)
After-Market Performance by Sector
Biopharma (N=18): 46.3% (average), 31.5% (median)
MedTech (N=23): (10.6%) (average), (12.8%) (median)
Source: CapIQ
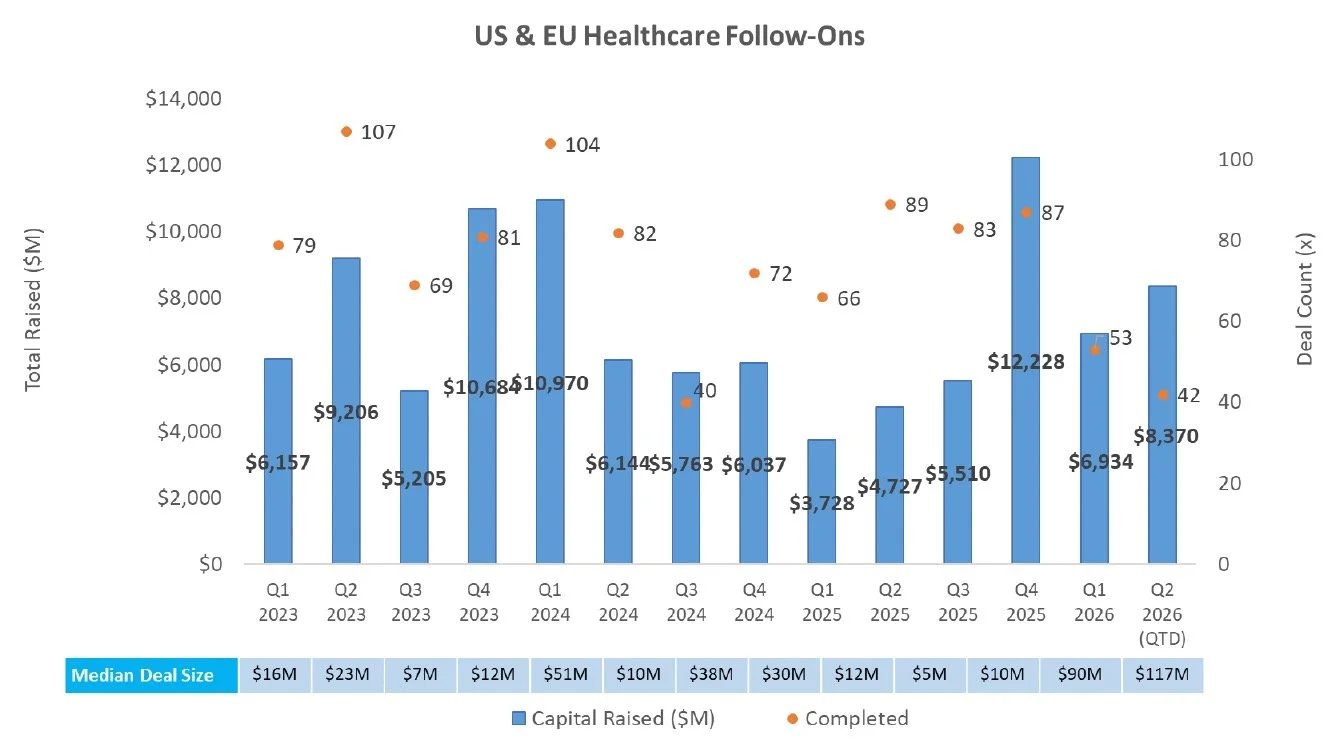
Follow On
There were two follow-on equity offerings totaling $11.5M last week
Source: Biomedtracker
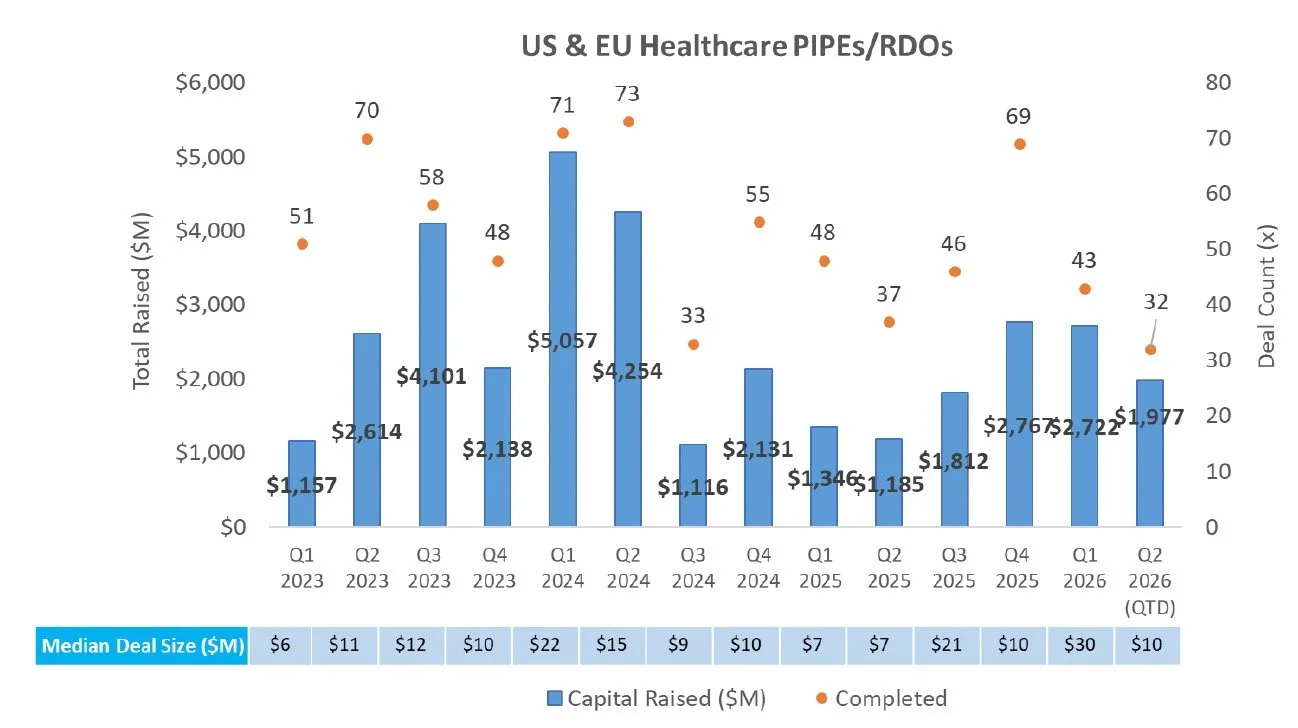
PIPE/RDO Markets:
There were five PIPEs/RDOs last week raising an aggregate of $96.0M
Whitehawk Therapeutics (NASDAQ: WHWK) announced an $87.5M PIPE financing to support working capital and general corporate purposes, including advancement of its antibody-drug conjugate (ADC) pipeline and related development activities, with proceeds expected to extend the Company’s cash runway into 2H 2028
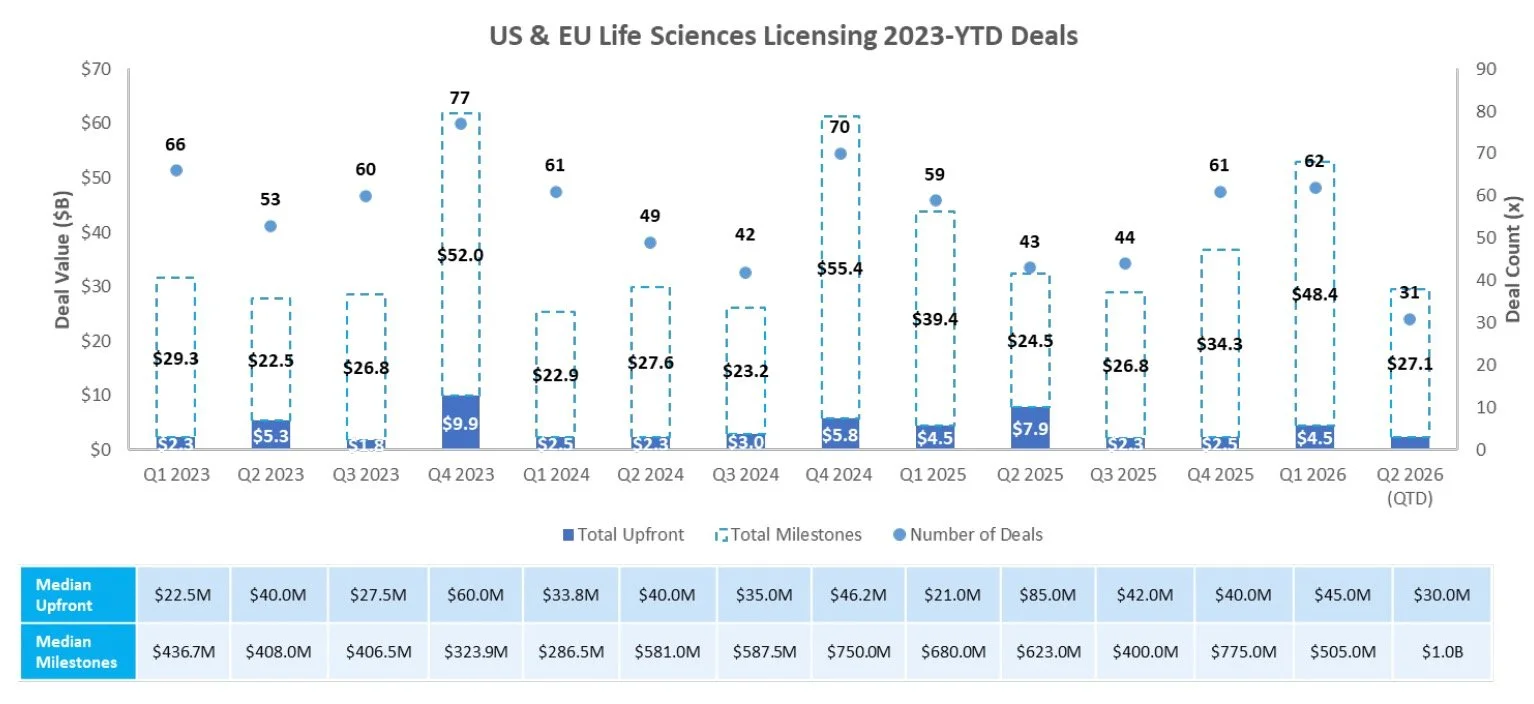
Licensing
Sources: Pitchbook, Biomedtracker, and CapIQ
Sources: Pitchbook, Biomedtracker, and CapIQ
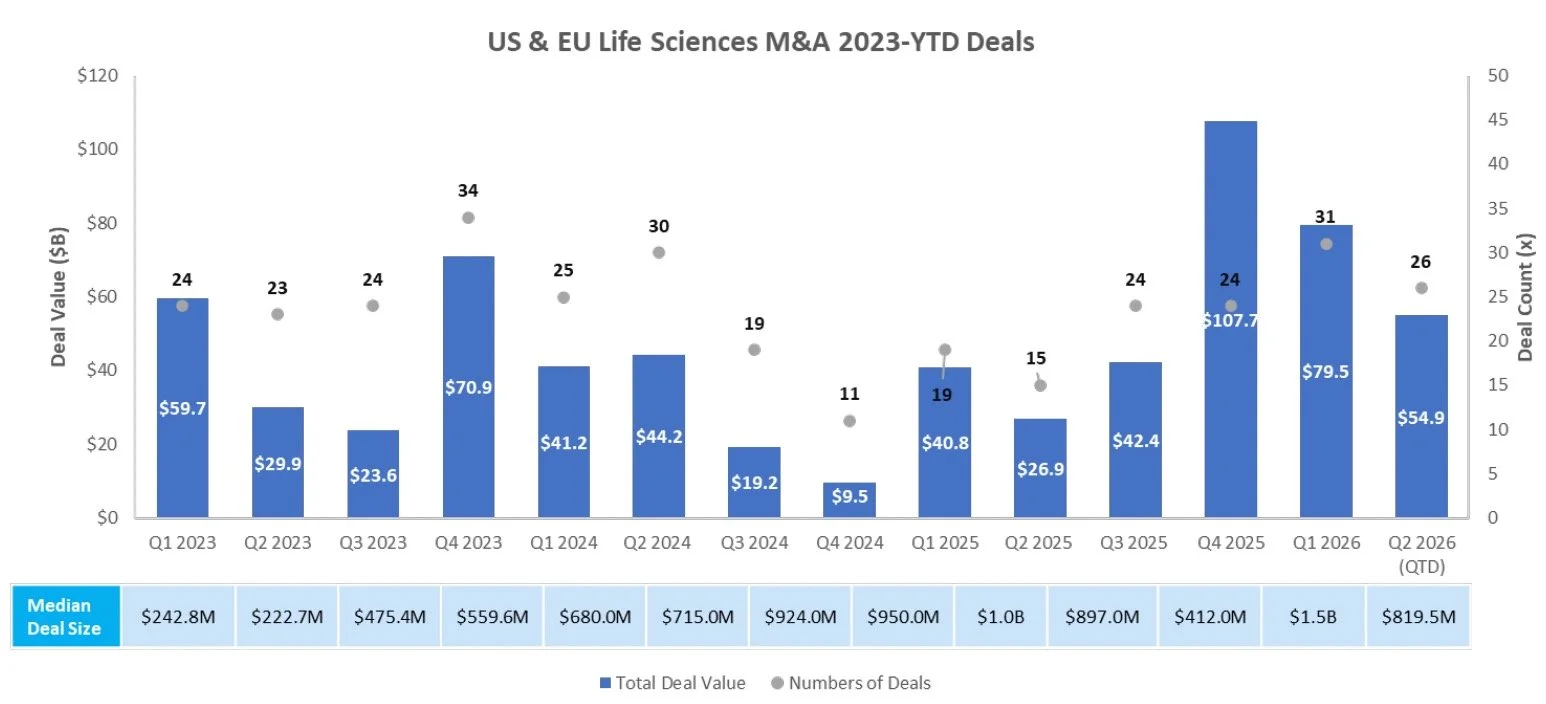
M & A
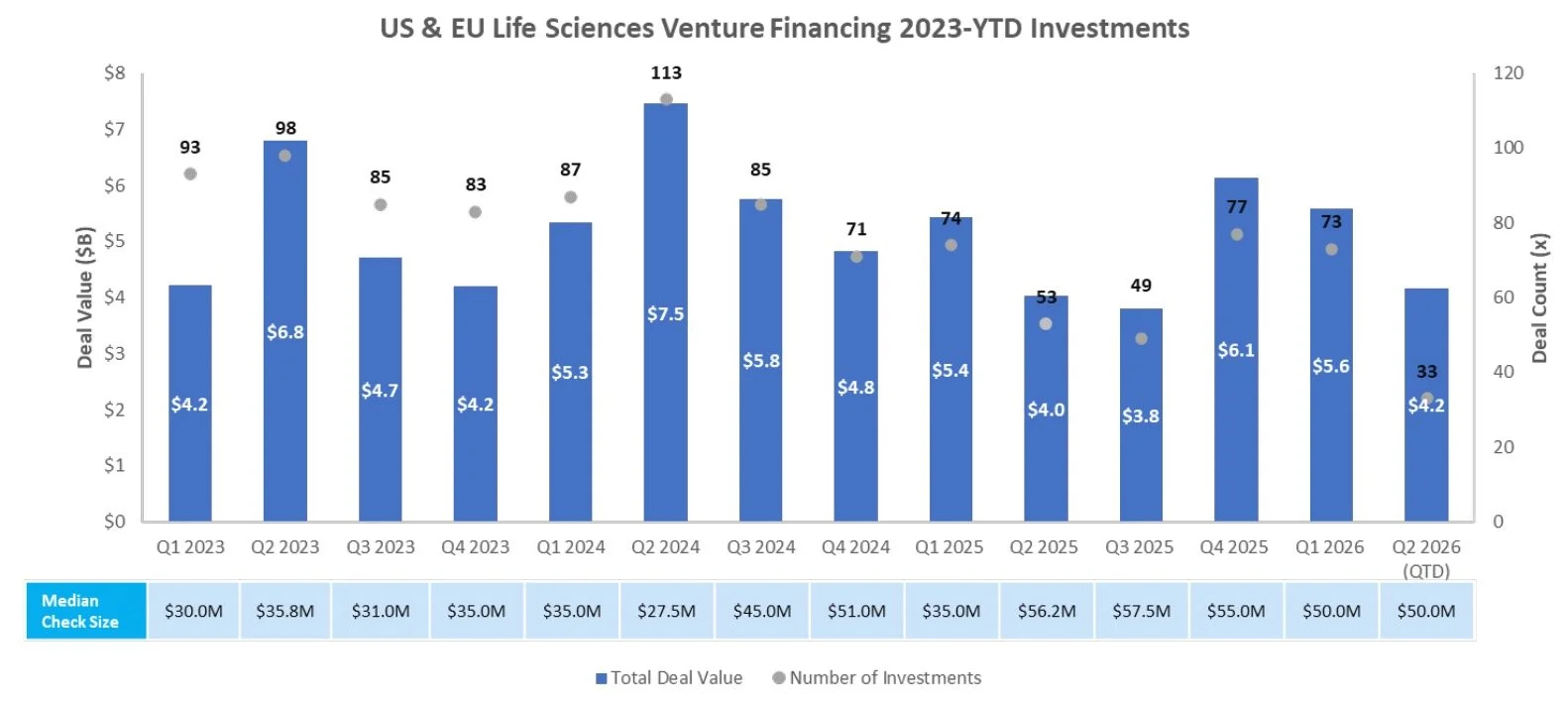
Venture Financing
Sources: Pitchbook, Biomedtracker, and CapIQ
CONNECT with the Authors >>
A MAP to the Future of Targeted Oncology
The latest in our series of healthcare analyst reports focuses on the mitogen-activated protein kinase (MAPK) pathway, one of the most commonly perturbed signaling pathways in human cancer. Flowing from RAS to RAF to MEK to EKR, the pathway is a master regulator of cell growth and survival. Therefore, the amplification of proteins or mutation of key signaling domains are a common hallmark of cancer.
HEALTHCARE MARKET REPORTS ARCHIVE
-
-
April 17, 2026
April 24, 2026 -
-
-
-
-
-
-
-
-
-
-
-
-
-
-
About DNB Carnegie | Back Bay
DNB Carnegie Back Bay drives global healthcare growth and innovation by providing a full range of strategic advisory and financing capabilities along the continuum of life science and healthcare company development. The DNB Carnegie Back Bay Healthcare Partnership is a marketing term referring to a strategic agreement between DNB Markets, Inc. and Back Bay Life Science Advisors. More information about the DNB Carnegie Back Bay Healthcare Partnership can be found here.
Securities products and services are offered in the U.S. through DNB Carnegie, Inc., a US-registered broker-dealer and a separately incorporated subsidiary of DNB Bank ASA. DNB Carnegie, Inc. is a member of the Financial Industry Regulatory Authority (“FINRA”) and the Securities Investor Protection Corporation (“SIPC”). Securities products and services are offered in the European Economic Area through DNB Carnegie.