10th Annual Nordic American Healthcare Conference Highlights and Key Takeaways
The 10th Annual Nordic American Healthcare Conference in New York City brought together healthcare companies, US healthcare investors, analysts, and industry leaders for two days of presentations and discussions focused on innovation, investment, and the forces shaping the market. Alongside presentations by 26 leading US and Nordic companies spanning biopharma, medtech, diagnostics, and health technology, DNB Carnegie Back Bay hosted expert talks and fireside chats on policy, regulation, AI, capital markets, and the future of drug development.
Back Bay’s key takeaways are highlighted below.
DAY 1: MARCH 25, 2026
-
Scott Gottlieb, MD, 23rd Commissioner of the U.S. Food and Drug Administration
Moderator: Jonathan P. Gertler, MD, CEO, Back Bay Life Science AdvisorsDr. Gottlieb addressed the current US regulatory environment, framing the central challenge for healthcare innovation as the slow erosion of trust, capacity, and predictability within the U.S. Food and Drug Administration (FDA) and Department of Health and Human Services (HHS). He noted that political rhetoric is loud, but long-term concerns include staff cuts—particularly of senior reviewers —fewer inspections, less engagement, and less expertise, which could make regulation less transparent and consistent over time, even if deadlines like PDUFA are met. He emphasized that modernizing regulations requires time and cannot be rushed by political cycles. For biotech, pharma, and medtech companies, this means operating amid uncertainty, where rigorous data, clear strategies, and strong leadership are vital. Ultimately, weakening stability could slow innovation, delay access, and reduce US competitiveness.
-
Speaker: Per Plotnikof, Vice President Corporate Communications, Investor Relations and Strategic Planning
Ticker: CPSE: ALK B
Country of origin: DenmarkDuring ALK’s presentation management noted a clear ambition to transform its long-established allergy franchise into a broader growth and innovation story, focused on disease-modifying treatments that rebuild tolerance over time, rather than just managing symptoms. The story is being told from a position of strength: DKK 6.3B in revenue for 2025, 15% growth in local currencies, a 26% EBIT margin, and 3.1M patients treated (up 500K from 2024).
Building on that foundation, ALK is expanding its Allergy+ platform into pediatric markets (ACARIZAX and ITULAZAX now in 21 and 12 markets, 4K+ prescribers), newer categories like neffy (DE, UK, and more markets in 2026), and peanut SLIT-tablet with Phase 2 topline data expected in Q2 2026. Strategic partnerships with GenSci, Torii/Shionogi, and ARS Pharma extend reach into China, Japan and beyond without outsized capital deployment.
The overall tone was confident and increasingly ambitious, with ALK framed as a profitable allergy leader leveraging strong cash flow to expand into adjacent categories, new geographies, and a more innovation-driven future.
-
Speaker: Thomas Feldthus, Chief Executive Officer
Ticker: Nasdaq Stockholm: SANION
Country of origin: DenmarkSaniona’s message was clear: they offer a differentiated pipeline, a more focused development plan, and the financial backing to support both. In 2025, the Acadia and Jazz deals, generating USD 70.5M in upfront payments, potential USD 1.6B in future milestones plus royalties, validated its ion channel platform and left Saniona with SEK 580.8M in cash. The tone was disciplined and confident, favoring a partnership-led model designed to generate meaningful non-dilutive funding while preserving strategic flexibility.
Clinically, Saniona is targeting two Phase 1 studies in 2026 and a third in early 2027 across SAN2668 (pediatric epilepsies), SAN2465 (MDD), and SAN2219 (epilepsy). Near-term partner catalysts add upside: Acadia’s Phase 2 in essential tremor, Jazz’s first-in-human study on SAN2355, and potential Medix royalties pending tesofensine approval in Mexico. The positioning is of a maturing CNS platform, capital-efficient, partnership-backed, and meaningful optionality across the pipeline.
-
Speaker: Michael Davidson, Chief Executive Officer
Ticker: NASDAQGM: NAMS
Country of origin: NetherlandsNewAmsterdam Pharma, a late-stage cardiovascular company aiming to revive CETP inhibition with a modern, commercially relevant profile, presented its positioning of obicetrapib as a differentiated, once-daily oral therapy that complements rather than competes with existing lipid-lowering treatments. Validation is accumulating with positive Phase [PB1] 3 data reported across BROADWAY, BROOKLYN, and TANDEM; a 2025 European marketing application by partner Menarini secured both monotherapy and fixed-dose combination with ezetimibe; and USD 729M in cash at year-end to fund and launch U.S. commercial buildout.
The broader ambition is a cardiometabolic franchise. BROADWAY delivered a 33% LDL-C reduction vs. placebo and exploratory 21% MACE reduction; PREVAIL (outcomes trial) has 9,500 patients enrolled; RUBENS launched in December 2025 for type 2 diabetes or metabolic syndrome; and an Alzheimer’s trial is planned for 2026 on the back of positive biomarker findings. The tone was ambitious yet grounded. This is not a single-asset lipid story.
-
Speaker: Kristoffer Nordström, Chief Financial Officer
Ticker: OM: XVIVO
Country of origin: SwedenXVIVO outlined a growth narrative driven by its goal to set a new standard in organ transplantation. Revenue remains diversified across Thoracic, Abdominal, and Services sectors, with a global footprint primarily in the Americas (55%), EMEA (39%), and Asia & Oceania (6%). The tone was mission-focused but also businesslike, emphasizing that XVIVO's mission goes beyond selling devices to addressing a core problem in transplantation, namely that many donated organs never reach patients.
Financially, XVIVO reported SEK 812.2M in net sales for 2025, SEK 88.4M in EBIT, with 2026 guidance of SEK 900M to SEK 1B in net sales and SEK 90M to SEK 150M EBIT, signaling a deliberate shift from expansion toward improved operational efficiency. Strategic priorities include commercial growth, expanded services, and R&D in heart and liver perfusion. The overall picture is a company using its expanding commercial and service base to advance transplant adoption, improve economics, and increase organ availability over time.
-
Speaker: Johan Luthman, Executive Vice President, R&D
Ticker: CPSE: HLUN B
Country of origin: DenmarkLundbeck, a global pharma company specialized in developing treatments for brain diseases, returned to NAHC with a noticeably sharper strategic focus and genuine commercial momentum. The company presented 2025 as another year of double-digit growth, led by Vyepti and Rexulti, with strategic brands generating DKK 19,011M, roughly 77% of total DKK 24,630M in revenue and DKK 5,275M in EBIT. The tone was confident and purposeful: Lundbeck is no longer just managing a mature neuroscience portfolio but actively reshaping around higher-growth brands and a more distinct pipeline.
The forward-looking story centered on pipeline transformation. Lundbeck expects five to six mid- to late-stage assets by the end of 2026, backed by positive Phase 2b results for bocunebart (migraine prevention), Phase 3 advancement of bexicaserin and amlenetug, and ahead-of-schedule enrollment completion MASCOT trial for MSA. For 2026, the company guides to 5% to 8% revenue growth at CER, 4% to 12% adjusted EBITDA growth at CER, and DKK 5.5B to DKK 5.9B in R&D spend, signaling a company willing to invest heavily in long-term neuroscience leadership while maintaining profitability.
-
Niels Granholm-Leth, DNB Carnegie Head of Equity Research, Denmark
Beth Percha, Chief Data and Analytics Officer, NY-Presbyterian Hospital and Adjunct Professor of Biomedical Informatics, Columbia UniversityColin Hill, Chief Executive Officer and Co-Founder, Aitia
Moderator: Jonathan P. Gertler, MD, Chief Executive Officer, Back Bay Life Science AdvisorsPanelists described AI in healthcare as a high-potential but unevenly adopted tool, with tension between practical utility and real-world complexity. Beth Percha noted that many health systems see AI as additive rather than time-saving, with physicians handling decision-making while facing more workflow, regulatory, and data-related concerns. She highlighted that deploying AI in complex care environments is difficult, especially when issues such as consent, compliance, and data use are not clearly addressed. Colin Hill and Niels Granholm-Leth pointed to AI’s long-term benefits in drug development, diagnostics, and innovation. Hill focused on AI’s role in uncovering further human biology and improving prediction, while Granholm-Leth emphasized AI’s value when it creates tangible end-user advantages. Overall, AI’s biggest impact will come from practical applications such as image analysis, registry review, and better-targeted development decisions.
-
Speaker: Laura Niklason, Founder, President and Chief Executive Officer
Ticker: NASDAQ: HUMA
Country of origin: United StatesHumacyte’s presentation highlighted a forward-looking story focused on broadening the narrative beyond a single commercial product, positioning Symvess as the initial proof point of a regenerative medicine platform, not the conclusion. Management connected current trauma indication and future opportunities in hemodialysis and coronary bypass, with a tone that was ambitious but execution focused.
USD 2.0M in 2025 revenue, 27 VAC approvals, 27 hospitals ordering, most with repeat orders, and a USD 1.475M Saudi Arabia purchase commitment signal early commercial traction. Key catalysts ahead: V012 Phase 3 hemodialysis topline data by June 2026, a supplemental BLA and first-in-human CABG study both targeted for H2 2026.
-
Speaker: Duane H. Barnes, President, Head of Sobi North America
Ticker: OM: SOBI
Country of origin: SwedenSobi’s presentation was disciplined and execution-focused, a company that has achieved scale and is now leveraging it for the next growth phase through commercial expansion, strategic investment, and pipeline progression.
Financially, Sobi reported SEK 28.238B in 2025 net sales, representing 15% growth at CER, and a 40% adjusted EBITA margin, with Q4 sales of SEK 7.821B, up 16% at CER. For 2026, the company guides low double-digit revenue growth at CER and adjusted EBITA margin in the mid-30s, with priorities including Altuvoct expansion, Aspaveli for nephrology, NASP for gout, Tryngolza pre-launch, Arthrosi integration, and Gamifant for IDS.
-
Speaker: Megan Richards, Vice President of Investor Relations
Ticker: OM: VICO
Country of origin: SwedenVicore presented with quiet conviction around a distinct scientific thesis in pulmonary fibrosis. Buloxibutid, a first-in-class oral AT2 receptor agonist with FDA Orphan Drug and Fast Track designations, is positioned as more than another IPF entrant.
Execution centers on ASPIRE, its global 52-week Phase 2b trial in IPF, with enrollment expanded from 270 to 360 patients and completion expected 1H 2026, backed by SEK 1,168.2M (~USD 127.0M) in cash. The tone was disciplined and data-focused, with the company approaching a meaningful inflection point from a position of financial strength.
-
Speaker: Fredrik Tiberg, President and Chief Executive Officer
Ticker: OM: CAMX
Country of origin: SwedenDuring their presentation, Camurus projected unusually strong operating credibility for a company of its size, reporting SEK 2,265M in 2025 revenues, SEK 933M profit before tax, and net cash position of ~SEK 3.7B. The tone was steady and execution-focused, not speculative.
The strategic focus is on converting that strength into the next growth phase. Key catalysts include a potential U.S. acromegaly launch (Oclaiz, June 10, 2026 PDUFA), SORENTO advancing neuroendocrine tumors, and CAM2056, its once-monthly semaglutide program, moving into Phase 2b study in 2026 following positive Phase 1b data.
-
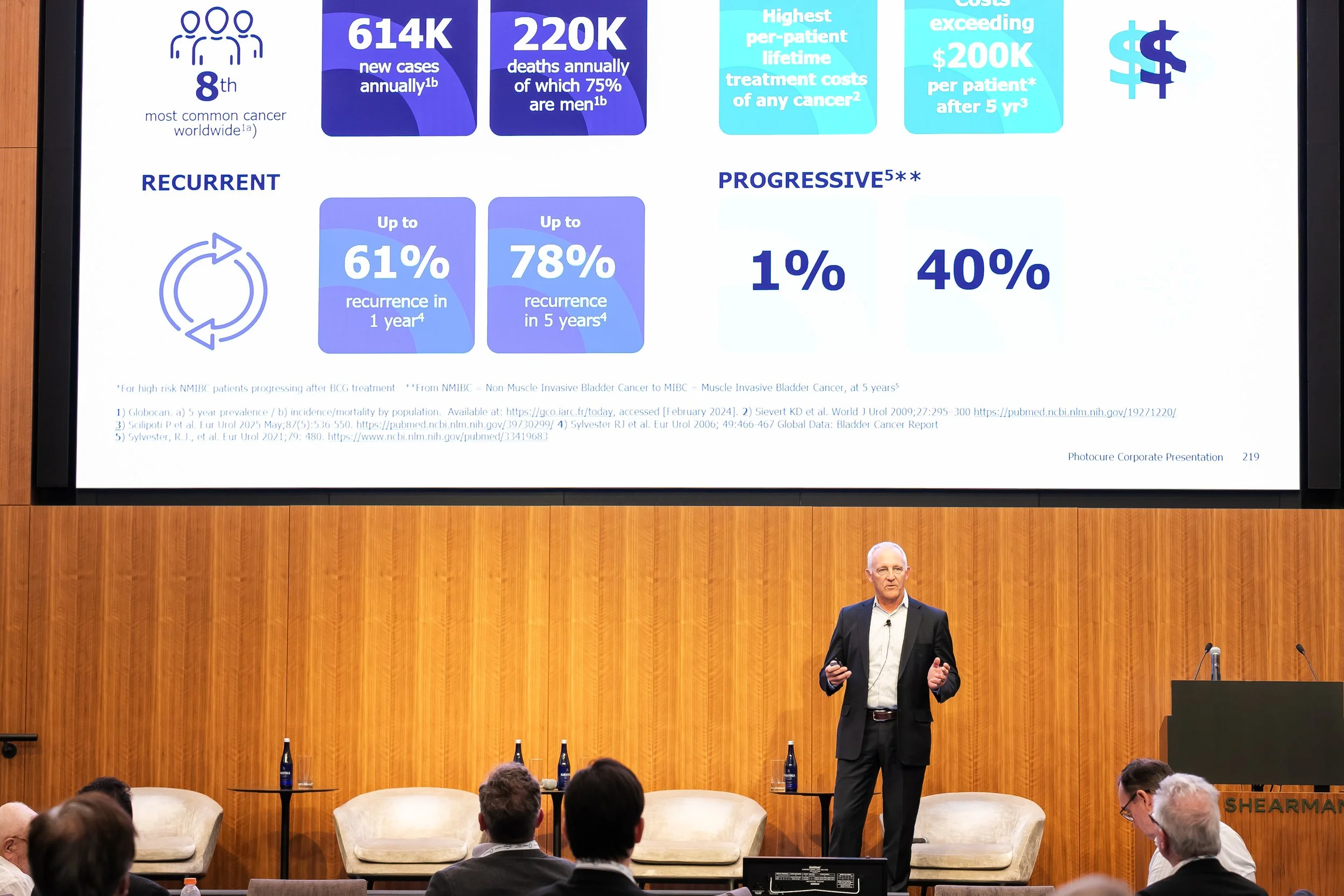
Speaker: Dan Schneider, President and Chief Executive Officer
Ticker: OB: PHO
Country of origin: NorwayPhotocure presented a confident, commercially focused growth thesis around Hexvix/Cysview with Blue Light Cystoscopy (BLC), a guideline-supported, clinically validated technology (300+ publications, 40+ prospective studies, 3,000+ U.S. registry patients) with management emphasizing that U.S. penetration remains low relative to the opportunity.
The strategy is to drive utilization within the installed base, expand access through tower upgrades and equipment partnerships, and shift toward precision diagnostics. NOK 530.1M in 2025 product revenues, NOK 46.2M in EBITDA, and 7% to 11% CER growth guidance for 2026 underpin a company moving from proving BLC's value to scaling market adoption.
-
Speaker: Kari Eian Krogstad, Chief Executive Officer
Ticker: MEDI.OL
Country of origin: NorwayMedistim presented itself as the global leader in transit-time flow measurement (TTFM) and intraoperative high-frequency ultrasound (HFUS), making the case that graft assessment can evolve from niche surgical tool to broader standard of care. The value proposition, immediate surgical feedback, reduced OR uncertainty, better decision-making, is backed by direct commercial presence across U.S., Canada, China, and key European markets.
2025 was a record year: NOK 699.8M in revenue (+24%) and NOK 196.2M in EBIT (+50%). Growth priorities include the newly launched MiraQ INTUI software platform to help drive combined flow-and-imaging procedures, expansion beyond CABG into vascular surgery, and clinical evidence generation through PATENT and ROMA-Women. The tone was confident and focused on execution, asserting that Medistim has significant growth potential in both well-established and emerging markets.
-
Speaker: Frederik Taylor Pitter, Investor Relations Director, North America
Ticker: CPSE: NOVO B
Country of origin: DenmarkDuring our discussion with Novo Nordisk, management acknowledged a more competitive obesity landscape while reinforcing its position as category leader with ~43% global GLP-1 volume share and DKK 82B in 2025 obesity care sales.
The near-term response is franchise broadening, not single-product defense. U.S. approval of Wegovy HD 7.2mg (March 19, 2026), U.S. launch of Wegovy pill (January 2026), and U.S. filing for CagriSema (December 2025). The tone was pragmatic and competitive, future growth will come from brand depth, broader access, and continued pipeline investment, not price-led
-
Speaker: Francesco Maria Lavino, Chief Executive Officer
Ticker: Private
Country of origin: US-UKDuring this presentation, F2G presented as a high-unmet-need innovation story with a clinical and medical affairs-first tone, built around olorofim, a novel oral antifungal candidate for invasive fungal infections where treatment options are limited or ineffective. The emphasis remained firmly on medical need and differentiated science, with the commercial opportunity reinforced by F2G’s validating partnership agreement with Shionogi for ex-US commercialization rights.
The company is in late-stage clinical development execution mode as it prepares for US commercialization. The 2023 FDA Complete Response Letter (CRL) remains in the rearview, seeming more like a regulatory step than a defining setback, with the full Study 32 dataset still central to the path forward and the global Phase 3 OASIS study ongoing. A USD 100M 2024 financing signals management is funding commercial readiness alongside development. The overall impression was focused and credible, though still higher risk than that of the more mature stories in the room.
-
Speaker: Paul J. Hadden, Senior Vice President, Investments and Business Development
Ticker: NASDAQ: LGND
Country of origin: United StatesLigand reported USD 161.0M in 2025 royalty revenue and USD 200M to USD 225M anticipated for 2026, with management emphasizing a more profitable and diversified business model.
The longer-term direction is compounding value through royalty growth, structured transactions, and portfolio diversification with expected long-term royalty receipts CAGR of at least 23% driven by programs including Filspari, Ohtuvayre, Capvaxive, Qarziba, and Zelsuvmi.
-
Speaker: Sveinn Sölvason, President and CEO
Ticker: CPSE: EMBLA
Country of origin: IcelandÖssur, now operating within Embla Medical, presented a confident, strategically grounded story centered on long-term leadership in mobility. The company reported USD 929M in 2025 net sales and 6% organic growth, with broad exposure across the Americas (43%), EMEA (49%), and APAC (8%), a business with real durability tied to structural demand in chronic mobility needs.
Growth is clearly concentrated in higher-end categories: Prosthetics & Neuro Orthotics grew 10% organically in 2025, while Patient Care grew 1% and Bracing & Supports declined 1%. The strategic direction is toward innovation-led mobility solutions, broader reimbursement, and neuro orthotics expansion, positioning Össur as a durable, patient-centric platform with a sales mix increasingly shaped by its highest-growth categories.
DAY 2: MARCH 26, 2026
-
James Cirenza, Managing Director, DNB Carnegie, Inc.
To open Day 2, James Cirenza focused on the market signals most directly influencing capital allocation decisions, especially the yield curve, issuance activity, and investor appetite for risk across healthcare and growth sectors. He pointed to the five-year and 30-year parts of the curve as key indicators of corporate financing conditions and broader economic health and suggested that while inflation and rate uncertainty continue to complicate the landscape, the market itself remains active. IPOs are still getting done, financing windows remain open, and the deals that are moving forward are, in many cases, getting larger.
He noted that healthcare may be better positioned than current sentiment suggests, particularly as investors remain heavily concentrated in large-cap technology. In his view, many investors are more exposed to tech than they realize, and smaller-cap healthcare and biotech could offer more relative value and better recovery potential than the market currently credits.
Overall, his message was measured but optimistic: this is a more selective market, not a closed one, and capital remains available for companies that are well-positioned and aligned with investor interests.
-
James Cirenza, Managing Director, DNB Carnegie, Inc.
Kristofer Liljeberg, Head of Healthcare Research, DNB Carnegie, Inc.
Bernhard Wu, Managing Director, Life Sciences, OMERS
Moderator: Gregory Benning, Managing Director, Head of Investment Banking, Back Bay Life Science AdvisorsThe discussion centered on a practical question: beyond the noise of policy changes, funding pressures, tariffs, and geopolitics, what truly influences healthcare investment decisions? James (Jim) Cirenza provided insight on how investors are balancing that uncertainty against the underlying value in healthcare, while Kristofer Liljeberg highlighted the pressure many Nordic medtech, device, and healthcare software companies face despite solid fundamentals, as U.S. pricing concerns, tariffs, and currency fluctuations continue to affect sentiment. Bernhard Wu offered a more stable long-term perspective, arguing that life sciences remain an attractive asset class because the demand for essential therapies and technologies does not vanish with short-term market fluctuations. He added that royalties and other non-dilutive structures continue to give investors a more resilient way to stay active in the space. Overall, the panel indicated that healthcare capital remains available, but is being allocated more cautiously, with greater emphasis on durability, selectivity, and long-term value.
-
Speaker: Tammi Gaskins, Chief Commercial Officer
Ticker: NASDAQ: IRWD
Country of origin: United StatesIronwood Pharmaceuticals presented Linzess as a durable commercial franchise that continues to matter strategically, pointing to 2025 Ironwood revenue of USD 296.2M, GAAP net income of USD 24.0M, adjusted EBITDA of USD 138.1M, and full-year 2025 U.S. Linzess net sales of USD 864.5M. The company also outlined a strong 2026 outlook, including U.S. Linzess net sales of USD 1.125B to USD 1.175B, total revenue of USD 450M to USD 475M, and adjusted EBITDA of more than USD 300M.
Beyond the financials, Ironwood highlighted apraglutide as its key pipeline asset, with key elements of the confirmatory Phase 3 trial in short bowel syndrome with intestinal failure finalized and site initiations expected to begin in the second quarter of 2026. The presentation showed that future growth will be supported by both the continued durability of Linzess and advancement of a late-stage rare disease program.
-
Speaker: Thomas Holm Pedersen, Chief Executive Officer
Ticker: Private
Country of origin: DenmarkNMD Pharma’s presented on a focused neuromuscular story, with ignaseclant positioned as the company’s lead asset and Charcot-Marie-Tooth disease emerging as the clearest near-term priority. In topline Phase 2a SYNAPSE-CMT results, the company said the exploratory study did not show a treatment difference on the pre-specified primary endpoint, the 6-minute walk test at 21 days, but did show improvement across multiple pre-specified secondary endpoints, including CMT-FOM, handgrip strength, fine hand function, and patient-reported outcomes.
Because NMD is privately held, the public figures are more focused on funding and program progression than quarterly financial reporting. The company says it has raised approximately USD 180M and continues to advance ignaseclant across CMT, generalized myasthenia gravis, and spinal muscular atrophy.
-
Speaker: Torbjörn Sköld, Chief Executive Officer; Håkan Johansson, Chief Financial Officer
Ticker: OM: BONEX
Country of origin: SwedenBONESUPPORT’s presentation centered on commercial momentum and scalability, around CERAMENT. The company reported SEK 1,174.7M in 2025 net sales (40% growth at CER), a 92% gross margin, and SEK 262.0M in adjusted operating profit.
CERAMENT was reinforced as both a product franchise and a broader strategic, a patented injectable bio-ceramic platform with drug-eluting capability. A key reimbursement milestone: CMS approval of NTAP for CERAMENT G in open fractures, effective October 1, 2025, providing up to USD 5,687.50 in additional reimbursement per case.
-
Speaker: Anders Hjort, Head of Investor Relations
Ticker: CPSE: AMBU B
Country of origin: DenmarkAmbu presented as a category leader in single-use endoscopy, with Endoscopy Solutions now the company’s primary growth engine, accounting for 60% of total revenue and delivering 15.4% organic growth in 2024/25. Full-year results: DKK 6,037M in revenue, 13.1% organic growth, and a 13.1% EBIT margin before special items.
Early 2025/26 momentum continues: Q1 Endoscopy Solutions grew 14.4% organically, overall organic growth was 8.6%, with full-year guidance maintained at 10% to 13% organic revenue growth and 12% to 14% EBIT margin. The tone was forward-looking, with emphasis on single-use conversion, commercial discipline, and platform differentiation through software and connectivity.
-
Speaker: Paul LaViolette, Chief Executive Officer and Co-Chairman of the Board of Directors
Ticker: NASDAQCM: PLSE
Country of origin: United StatesPulse Biosciences delivered a growth story centered on next-generation pulsed field ablation, with atrial fibrillation positioned as the lead clinical and commercial opportunity. Financials reflect an early-stage company: USD 350K in 2025 revenue, a USD 72.8M GAAP net loss, and USD 80.7M in cash at year-end.
Clinical momentum is building: 100% procedural success at 6 months and 96% at one year in evaluable patients from the AF feasibility study, FDA approval for a pivotal IDE study in paroxysmal AF, and ongoing enrollment in NANOCLAMP AF and PRECISE-BTN (thyroid). The message was ambitious: near-term value creation comes from late-stage cardiac execution, with broader platform potential beyond electrophysiology preserved.
-
Speaker: Martin Welschof, Chief Executive Officer
Ticker: Nasdaq Stockholm: BINV
Country of origin: SwedenBioInvent has a focused immuno-oncology strategy built around two lead antibody programs, BI-1808 and BI-1206. Financially, the company reported SEK 226.5M in 2025 net sales, a SEK -332.9M loss after tax, SEK -247.8M in operating cash flow, and SEK 592.7M in liquid funds, alongside strategic portfolio changes intended to accelerate the lead programs and enhance value creation.
Beyond the financials, BioInvent reported promising BI-1808 data in recurrent ovarian cancer, including a 24% overall response rate in combination with pembrolizumab, and said the ovarian cohort expansion is expected to read out in H2 2026. It also highlighted BI-1808 monotherapy data in CTCL, showing a 46% objective response rate and 92% disease control rate in 13 evaluable patients, while continuing to advance BI-1206 in NHL and in a Phase 2a study in treatment-naïve NSCLC and uveal melanoma.
-
Erik Hultgard, Senior Healthcare Analyst, DNB Carnegie
Ester Caffarel-Salvador, Director, Strategic Innovation, Global Rare Diseases, Chiesi
Elaine Jones, Chair, BOOST Pharma
Moderator: Stephan Gauldie, Managing Director, Back Bay Life Science AdvisorsThis discussion focused on how investors evaluate complex therapeutic platforms, where the opportunity is significant, but the risk profile is multi-layered. Panelists noted that drug development now requires looking far beyond scientific promise to include manufacturing, scale-up, distribution, payment, regulatory pathways, and clinical execution. Erik Hultgård emphasized the difficulty of valuing platform technologies in public markets, particularly given modality-specific hurdles such as cell-therapy logistics or radiopharma infrastructure. Ester Caffarel-Salvador pointed out that in rare diseases, strong efficacy, safety, a credible FDA pathway, and clear inflection points are especially important, especially in cautious environments. Elaine Jones reaffirmed that investors seek platform potential combined with a smart strategy, multiple targets, and a clear route to value. Overall, companies need a disciplined plan to fund development through meaningful data, minimize risks, and achieve measurable milestones.
-
Speaker: Tuukka Hirvonen, Head of Investor Relations
Ticker: HLSE: ORNBV
Country of origin: FinlandA diversified Nordic pharmaceutical company founded in 1917, Orion Pharma operates in more than 35 countries, with R&D efforts centered on oncology and pain. Its main message highlights that innovative medicines and Nubeqa are fueling growth, while branded products, generics, consumer health, animal health, and fermion offer stability and strong cash flow.
Orion reported 2025 net sales of EUR 1,889.5M (+22.5%), and an operating profit of EUR 631.6M (+51.6%); R&D spend grew to EUR 210.4 million (+17.2%). The company noted Q4 was boosted by a EUR 180M milestone related to Nubeqa, and its pipeline includes late-stage and mid-stage programs covering darolutamide, opevesostat, levosimendan, ODM-212, and several biologics candidates, with a particularly busy readout schedule anticipated from 2026 to 2028.
-
Speaker: Pär Ihrskog, Chief Financial Officer
Ticker: OM: VITR
Country of origin: SwedenVitrolife presented as a leading reproductive health platform, helping fertility clinics standardize and improve IVF workflows across five strategic pillars: owning the platform, innovating to strengthen leadership, accelerating growth in key markets, optimizing the go-to-market strategy, and driving operational excellence. The broader market is compelling: infertility affects 1 in 6 people worldwide, less than 1% of babies are born via IVF, estimated IVF cycle growth is about 5%, and successful treatment outcomes are roughly 33%.
Vitrolife reported SEK 3,440M in 2025 (4% organic growth in local currencies and a 5% decline in SEK due to currency headwinds) and SEK 949M EBITDA (27.6% margin). Revenue mix is well balanced, with Consumables (40%), Genetics (40%), and Technologies (20%). Long-term targets: 10%+ organic growth in local currencies by 2028, a 33% EBITDA margin, and net debt/EBITDA ratio of 3x.
-
Speaker: Fredrik Dalborg, Chief Executive Officer
Ticker: OM: ALIF B
Country of origin: SwedenA top independent European life sciences partner, AddLife operates in 30 countries with 2,300 employees, 54,000 customers, and 3,500 suppliers. The strategy relies on strong market positions, operational flexibility, and acquisition-led growth, with a focus on advanced products, digital solutions, and trusted local service organizations.
Financially, AddLife reported SEK 10,442M in 2025 net sales, SEK 1,417M in EBITA, and a 13.6% EBITA margin. Profitability (P/WC) came in at 62%, well above the 45% threshold, and net debt/EBITA of 2.2 beat the 3.0 target. The company reaffirmed its goal to double EBITA within five years (15% avg annual growth via organic and M&A), with three acquisitions completed in 2025: Edge Medical, Pharmacold, and Opitek.
-
Speaker: Theresa Agnew, Chief Executive Officer and President
Ticker: BIOG B
Country of origin: SwedenBioGaia presented as a science-driven probiotics company with clear ambition: become the most trusted probiotics brand worldwide. The strategy centers on three pillars: grow the core, expand through direct markets, and drive breakthrough innovation. The company established direct distribution in France and the Netherlands in 2025, with Germany and Austria planned for early 2026.
BioGaia reported SEK 1,538.2M in 2025 net sales (14% organic growth), with Pediatrics contributing SEK 1,154M and Adult Health SEK 372.5M. The U.S. was a standout with SEK 316M in record sales, up 30% organically. The company backs its credibility with ~290 clinical studies, 25,000+ participants, and ~10% of net sales reinvested in R&D, with 40% of sales now originating from its own subsidiaries.
-
Katrina Armstrong, MD, Executive Vice President for Health and Biomedical Sciences, Columbia University
Moderator: Jonathan P. Gertler, MD, CEO, Back Bay Life Science AdvisorsDr. Katrina Armstrong’s discussion focused on a fundamental gap in healthcare: the disconnect between what science can achieve and what patients experience. She stressed that improving outcomes involves considering the full context of a patient’s life, including access barriers, trust, care delivery, and social factors that influence whether innovations and advances reach those in need. She referenced breast and prostate cancer examples to show disparities in care are often driven less by biology and more by referral patterns, provider choices, and whether the system aligns with the patient’s values. A central theme was trust. Dr. Armstrong argued that trust is built on competence and shared values. She emphasized that healthcare institutions often focus too much on expertise and not enough on demonstrating that patients’ priorities come first. She also highlighted the widening gap between where innovation occurs and where patients receive care, but expressed optimism about faster translation of ideas, personalized medicine, mental health care, and living medicines. Her overall message was that science alone isn’t enough; delivery, access, communication, and trust are vital for meaningful change.
About the Nordic-American Healthcare Conference
The Nordic-American Healthcare Conference (NAHC) is the leading US-Nordic healthcare equity conference in the US with a sole focus on US-Nordic collaboration and healthcare growth.
Now celebrating its tenth year, the NAHC continues to unite innovative life science companies with exceptional technologies or advances from across the Nordic region and North America with healthcare investors, industry leaders and entrepreneurs for two days of strategic dialogue, company presentations, company-investor meetings and networking.